When chemotherapy drugs leak from the veins it can cause serious injury to the patient, greatly heighten their fears of undergoing future treatment cycles, and undermine their trust in their medical team. Knowing how to assess and reduce the risks, and what to do when things go wrong, is essential.
Extravasation occurs when medicines leak from the compartment where they are intended to be, such as a vein or muscle, into the surrounding tissues. The impact of this accidental administration depends on the pH and metabolic effects of the drug on the tissue. Some drugs cause no harm while others can cause serious injury such as loss of function or tissue damage requiring grafting or, in extreme cases, amputation.
Key factors to consider in recognising and reducing the risk of extravasation are:
- the patient to whom you are giving the drug
- the drug that you are giving and its potential for causing harm if extravasation occurs
- the device you are using for administering the drug
- any risks that may be associated, and how to mitigate them
- observation of the patient while they are having the infusion and after an extravasation injury.
Patient factors
When we consider the patient, we need to think about the patient’s previous experiences, including use of parenteral therapies, which will impact on their preconceptions and also their vein access. Medical history must be considered, because patients with peripheral vascular disease, neuropathy, stroke and diabetes have specific requirements, and these conditions can increase the permeability of the veins and increase the risk of extravasation. Patients who have neuropathy or have had a stroke may not sense any changes, so if you cannulate an area of numbness they may not be aware or able to tell you there is something wrong until the injury is quite extensive.
We also need to consider how well a patient can communicate. Children may not have the vocabulary to tell you that something’s wrong, and patients with impairments affecting communication may not be able to tell you that things aren’t right, so you need to observe them carefully and recognise changes in their condition.
It’s important to empower patients to be partners in their care and encourage them to communicate any concerns that they have. My experience has been that there are patients who tell me if the area around where their chemotherapy is being given hurts, or is sore, or numb or painful. However, some will not mention these symptoms. It is helpful to ask patients whether the area feels different, which may give you more objective feedback.
Patients should be made aware of the risk of extravasation. Before we start treatment we tell patients the basic facts, without going into unnecessary detail. This includes explaining that, on occasion, the cannula or lines may leak and this may require urgent intervention depending on the drug being infused.
Drug factors
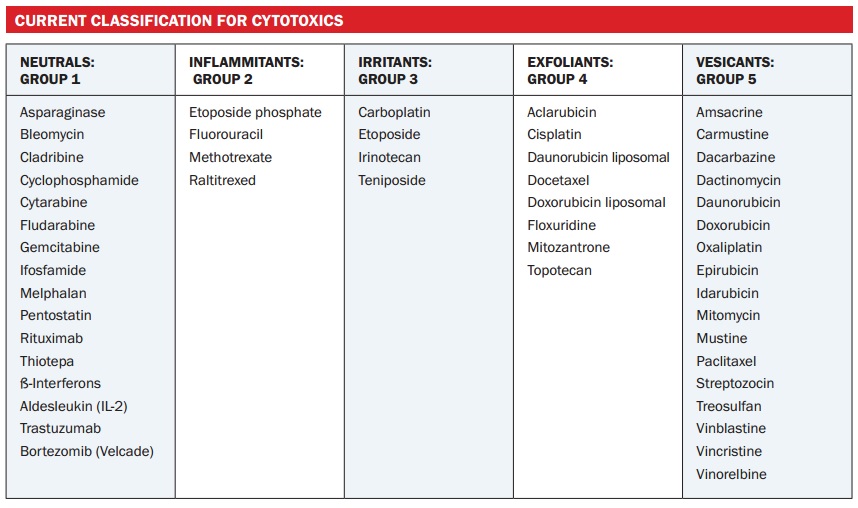
In the UK we use the five-point grading system for drugs in terms of the tissue damage they cause:
- Vesicant – blistering and necrosis
- Exfoliant – inflammation and skin shedding
- Irritant – sclerosis, burning, local warmth, hyper-pigmentation, discomfort, erythema or tenderness n
- Inflammitant – flare, inflammatory reaction
- Neutral – no inflammation or tissue damage on extravasation.
Across Europe, the categories of vesicant and exfoliant are often put together. The table opposite classifies commonly used cytotoxics into the five categories. The vinca alkaloids and the anthracyclines are groups of agents that are frequently used, and we need to be aware that they can cause significant tissue injury should they extravasate.
We tend to associate extravasation with cytotoxic agents, but other drugs and agents, such as sodium bicarbonate, hypertonic saline, and diazepam, can also act as vesicants.
Question:Who should be responsible for assessing patients for extravasation risk before their therapy starts?
Answer:This varies in different countries. Doctors give infusions in some countries, but in the UK most chemotherapy infusions are administered by nurses. My personal view is that the person who is going to administer the chemotherapy should assess the patient. This should include assessing the quality of the patient’s veins, considering the risk of extravasation at different infusion sites, as well as medical history and conditions such as diabetes or stroke.
Question:We know how busy everyone is, but how much time do you consider necessary for a basic assessment of a patient?
Answer:Everybody is pushed for time, but there is a risk of overlooking something important if you have not fully assessed the patient and relevant risk factors before treatment, which increases the risk of problems. Assessing the patient’s medical history, including any conditions they have and treatments they are on, and other factors influencing the patient, takes a few minutes, plus a couple of minutes observing them in the clinic. Vein assessment takes a little longer, probably 15–20 minutes
Choice of device
The choice of device is very important. I am mindful of the fact that in the UK we have access to virtually any device we want to use to gain intravenous (IV) access, but some countries do not have the opportunity of using an implanted port or a peripherally inserted central catheter (PICC) line, and so they have to use an alternative option. However, where you know you are using highrisk drugs that could potentially cause harm, the use of a central line allows large volumes to be infused into a large vein, achieving rapid dilution of the drug (see figure below). Another advantage is that a central line can be buried deep under the skin. The devices that tend to be used for central access are Hickman lines and PICC lines, or you can use an implanted port (Portacath).
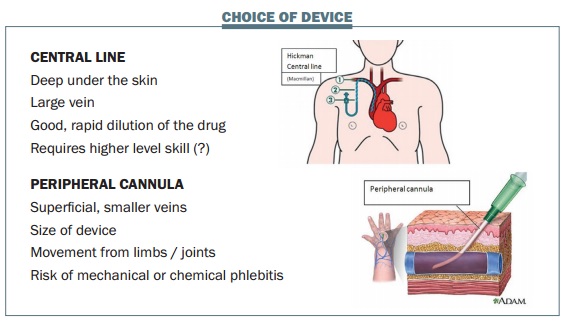
Peripheral cannulas are more commonly used than central lines, partly based on cost. They are used in superficial veins, which tend to be smaller, so it is necessary to consider the size of the device that is going into the vein. In the past, a large device might be used to give a large volume, but a very small device can still deliver a large amount of fluid. The small yellow 24-gauge cannula is often called a paediatric or neonatal cannula. Despite having a very narrow lumen, they can infuse 22 mL per minute, which amounts to 220 mL in 10 minutes, and it is unlikely that a faster rate would be required.
Small cannulas leave only a small puncture hole when removed, reducing the risk of extravasation. In addition, you can draw blood from one of these small lines, although drawing too quickly through such a narrow lumen can haemolyse the sample.
Most people are skilled in the use of a peripheral cannula, while a higher level of skill is required for using a central line. But inserting a peripheral cannula in the correct location is very important. If a cannula is inserted near the wrist or another joint where there is going to be a lot of movement, this can cause friction within the vessel, which can lead to inflammation in the vessel wall (mechanical phlebitis). In a permeable vein this movement can encourage infiltration through the vessel wall and transport the drug into the surrounding tissue. Ensuring a good flow of blood around a small cannula will allow the drug to be diluted quicker and also minimise the risk of direct chemical contact with the vessel wall, so reducing the risk of chemical phlebitis.
Assessing the patient’s veins is also essential in selecting the correct device. This should include consideration of the duration of therapy and the total number of vein accesses required to complete treatment and the frequency of infusions. Device choice should also take into account the drug to be infused and the patient’s preference. A central line may be more appropriate where the veins are not very good, but this also depends on the number of cycles that are going to be infused and infusion time.
The drugs that are going to be infused are important to consider, because extravasation from a peripheral cannula may lead to a significant impact on the function of the affected limb. For this reason we avoid the back of the hand and the anticubital fossa, because of the major impact of tissue damage on functioning.
We also have to consider patient choice. Some patients say that they don’t want a central line and we should honour their choice, but let them know that, if the veins become impossible to access peripherally, they may need a central line. The cost of using a central line versus the benefits to the patient has to be considered as well. Some lines cost hundreds of euros to place, whereas peripheral cannulas may be a few euros. The benefit to the patient of having a peripheral cannula is that it comes out after treatment, and they can get on with their life without any equipment requiring further care. A central line is the easiest option for healthcare professionals to access, provided that there are no problems with the line, and each treatment may be quicker.
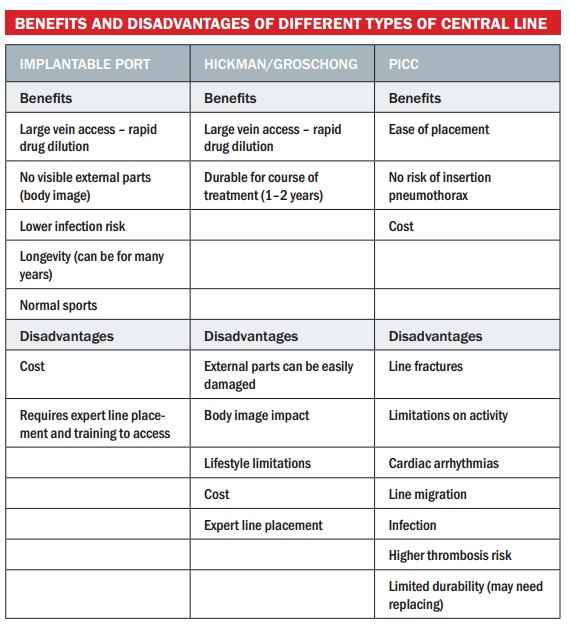
The table below summarises the benefits and disadvantages of different types of central lines. Implantable ports are placed in large veins, achieving rapid dilution of the drug. They don’t usually have any external visible parts, which reduces risk of infection, and it has good longevity, lasting up to 10 years. Patients like this option because they don’t have external parts to deal with, body image may be more positive and they can play sports. However, it is the most expensive form of central line and requires expert line placement and training to access. The Hickman or Groschong line also accesses large veins and achieves rapid drug dilution with good longevity. However, there are external parts that can be damaged and also affect body image. They are more costly than peripheral cannulas but less expensive than implantable ports. The PICC line is easy to place, with no risk of insertion pneumothorax and is cheaper than the other two options. Disadvantages include line fracture, limitations on activity and limited durability.
Peripheral cannulation requires good veins that will last the duration of the patient’s treatment course. A cannula is suitable for short courses of treatment in patients with healthy veins. It can be quite cost-effective, is quick and easy to place and is removed after use. There is a good range of size available, with the 24-gauge (yellow, with colours being universal) being excellent for giving peripheral IV drugs. In my experience it’s very rare that you need to use a bigger cannula for routine infusion. The choice of device is very important, selecting the smallest to deliver the drug over the time required. This also gives faster dilution of the drug, as the blood flows past the cannula and allows dilution along the way. Minimising the chemical contact with the vessel wall reduces the risk of extravasation injury occurring.
Disadvantages of peripheral cannulation include the fact that the wrong device is often chosen. Staff are often unaware that a smaller device is better. It also requires expertise to place and choose the insertion site correctly, and is not suitable for long courses of treatment if patients have poor veins.
Selecting the vein, I tend to start with the patient’s non-dominant arm, using the most distal site suitable for initial cannulation wherever possible, but avoiding the back of the hand, moving higher up the arm with any subsequent attempts to avoid leakage and extravasation. Subsequent cannulation attempts should be at a site proximal to the initial insertion. Cannulation attempts should be avoided on skin with altered sensation or on skin that is bruised, painful or infected.
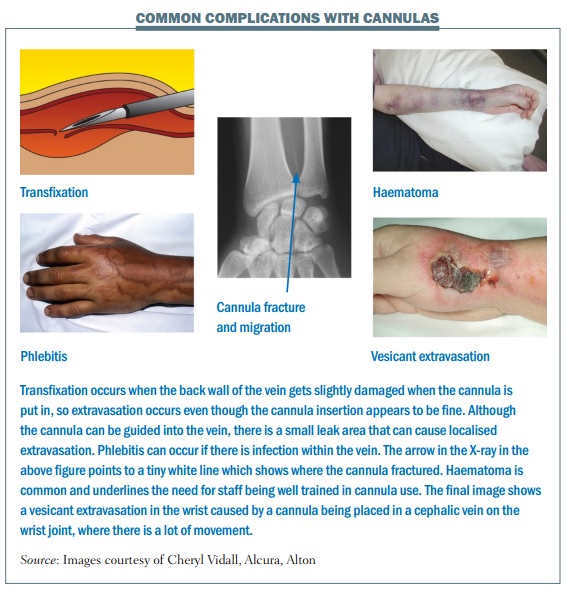
The figure below shows examples of common complications with cannulas, which can include:
- Mechanical phlebitis due to the device rubbing on the vessel wall
- Chemical phlebitis caused by the drug making contact with the vessel wall
- Thrombophlebitis
- Extravasation or ‘tissuing’
- Infection introduced through poor cannulation technique, contamination of drugs or skin contact
- Infusion reaction
- Embolism, from the drug, glass or thrombus formation
- Migration, mislocation, fracture, or the cannula falls out.
Preventing complications after the cannula insertion
Asepsis is essential, using non-touch technique throughout. It is important to monitor the site throughout the infusion and avoid over-manipulation of the cannula. All connections should be secure to avoid drugs leaking out, and it is essential to ensure the drip does not run dry. A good IV dressing should be used to hold the cannula stable and minimise the risk of movement while the cannula is in the vein. The infusion rate and the dilution of the drug are very important in preventing complications. The drug concentration can cause irritation to the vessel and the pH can be affected by the amount of diluent. What you want is fast dilution of the drug to minimise the risk of chemical injury.
The risks of extravasation
Extravasation causes pain, injury and loss of function to the patient. It can also mean delays in treatment for cancer, which can reduce efficacy and may lead to fear of having further cycles. It may also mean having to rebuild confidence in the clinical team. Nurses often feel responsible for the injury having occurred, and it may reduce their confidence in giving chemotherapy. The problem can also lead to loss of reputation from an organisational perspective, and may result in compensation claims.
Measures to reduce the risk of extravasation
It is essential to observe patients and monitor what is going on during treatment. Ask patients to report any change in sensation without delay. If using a pump or monitor, make sure the pressure sensors are set very low, because a change in pressure could suggest that something is going on either within the device or the vein. Observe the flow rate and take any resistance seriously.
The position, size and age of the venepuncture site are the most important factors to consider in the prevention of extravasation. The risk can be significantly reduced by the following measures:
- Use a central line or peripherally inserted central catheter (PICC) for slow infusion of high-risk drugs
- Never leave a butterfly if administering cytotoxic drugs; stay with the patient throughout (or avoid butterflies altogether)
- Avoid small and fragile veins
- Use a recently sited cannula Site the cannula so it cannot be dislodged
- Use the forearm and avoid sites near joints.
Managing extravasation
Immediate action is essential when extravasation occurs. Aspirate the cannula and then remove it and document the amount drawn back from the aspiration, even if this is zero. Palliate the patient’s immediate symptoms with cold or warm analgesia (depending on the drug). Mark the affected area, take a photo and make sure the clinical team is aware of what has happened. Treat the patient with Savene for anthracycline injuries (or dimethyl sulfoxide [DMSO] 99% if no Savene), and check the protocol for other injuries.
The ESMO–EONS guidelines on the ‘Management of Chemotherapy Extravasation’ (Ann Oncol2012, 23 suppl 7:vii167-vii173) provide a lot of information and the best management practice.
Summary
Extravasation is a risk with chemotherapy and other drugs given intravenously. It is essential to consider the patient, what drug is being given, and which device is being used in order to assess and minimise the risk of extravasation. Patients should be observed and monitored carefully during administration of chemotherapy, being alert to any change in sensation. We need to document care and observations together with actions and follow-up when extravasation occurs, and involve the patient in their treatment plan.






Leave a Reply