The more we learn about biological variations and changes within a tumour the more daunting becomes the challenge of personalising therapies.
On the road towards personalised cancer therapies, the tasks of identifying new targets and devising ways to hit them seem to be coming along quite nicely. Right now, the big challenge is all about finding ways to work out which of the rapidly expanding selection of therapies will work best for the patient in front of you. Clinicians are crying out for validated cost-effective and patient-friendly methods for gathering biological information (‘biomarkers’) that help them to select the most appropriate therapy option.
Some of these biomarkers are already well known – the FISH test for HER2 amplification predicts response to therapies designed to block HER2 signalling, such as trastuzumab or lapatinib, while KRAS mutation is a marker predicting resistance to EGFR inhibitors such as cetuximab and panitumumab. And long before these, oncologists were using levels of oestrogen receptor and progesterone receptor as markers for response to hormonal therapy, for instance in breast cancer.
Progress can be seen in the way that pathology labs are introducing an increasing number of tests for biomarkers into the diagnostic routine. Outside the hospital setting, a whole diagnostics industry is mushrooming to provide testing kits to hospital labs and to offer diagnostic services for more high-tech tests. Examples include Genomic Health’s Oncotype Dx multi-gene assays and the Agendia and Affymetrix genomic microarrays, which can be used to help select patients for adjuvant chemotherapy– technologies developed initially for use in breast cancer, but now introduced across a variety of cancers.
Progress is also taking place in an area not so well known to the oncology community. The next big thing in biomarkers may be all about ‘functional’ imaging, which tells you not what a tumour looks like, but about what it is up to biologically. Two imaging technologies in particular are exciting interest for the potential they offer to help inform clinical decision making. The most surprising, perhaps, is MRI. Valued for decades for its ability to provide anatomic images of soft tissue lesions, this technique turns out also to have potential for imaging tumour microenvironments and cell metabolism – necrosis, cell density, metabolism, tissue perfusion and oxygenation. This can provide vital information about the nature of a tumour and how it is responding, or likely to respond, to a given therapy.
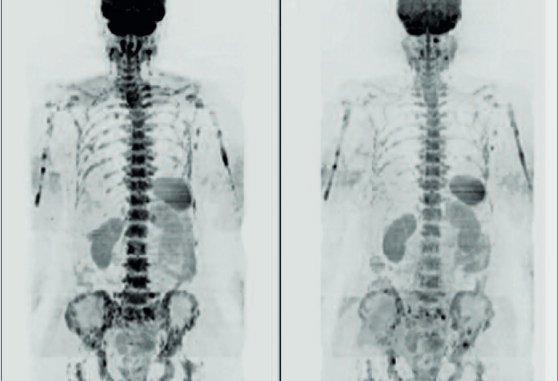
The other technique of interest comes from the field of nuclear medicine, in the form of PET (positron emission tomography) or SPECT (single-photon emission computed tomography). These techniques make it possible to visualise how an injected substance moves around the body by ‘labelling’ it with a tiny amount of radioactive tracer.
Oncologists will be familiar with the increasing use of FDG-PET for measuring response to treatment, particularly cytostatic treatments of solid tumours, where response typically does not take the form of tumour shrinkage, with the result that traditional anatomical imaging using CT or MRI can be misleading. This PET procedure uses FDG, a glucose analogue labelled with a fluorine radio isotope (18F-fluorodeoxyglucose), to map levels of glucose uptake around the body. This is in the process of being validated as a RECIST (Response Evaluation Criteria In Solid Tumours) marker of early response.
Glucose uptake – a generic marker of tumour activity – is only one of a number of markers of interest for clinical decision makers. The PET technique, in theory at least, can be adapted to map any biological process or molecular marker that can be delineated by a labelled compound that can safely be used in a patient. This includes specific targets such as oestrogen, HER2 or EGF receptors, as well as more generic biological markers of hypoxia, cell proliferation, and cell death.
A new generation of PET-MRI scanners has addressed many of the technical and practical challenges of functional imaging. The question now needs to be answered for both PET and MRI: in what way can they contribute to the everyday practice of personalised cancer therapies?
An alien in the imaging world
Elisabeth de Vries is a professor of medical oncology at the University Medical Centre Groningen, in the Netherlands. So convinced is she of the potential value of imaging for clinical decision making that she has waded in as “an alien in the imaging world”. Her recent research efforts have focused on investigating the clinical use of PET/SPECT, and more recently fluorescence imaging.
de Vries believes these imaging techniques offer a way to address some of the knottiest problems in personalising cancer therapies – not least, the growing recognition that the biology of a tumour can vary markedly from one area to the next and metastatic lesions do not necessarily resemble the primary tumour. “We all want to move to personalised medicine. We want to know who needs what drug either before or early during treatment. But one of the things that I find remarkable is this heterogeneity in tumour lesions. Tumour biopsies provide only static information on the status of a marker in a small part of the tumour and disregard the remaining tumour and possible metastases. Imaging can give us a better wholebody picture and insight into all lesions.” What’s more, she adds, because it is noninvasive it can be used repeatedly.
Access to this level of information can be particularly important in cases where standard diagnostic tests are giving conflicting information, says de Vries. “You have a patient with two breast cancers, for instance – one on the right and one on the left. This patient develops metastases, and they are hard to biopsy. You know one primary is oestrogen receptor positive and the other is not. If you can do a PET scan using 18F-oestradiol [FES], which binds to the oestrogen receptor, you can confirm whether or not ER is present on the metastasis” – potentially important information when it comes to choosing a therapy.
de Vries is setting up a prospective study in three Dutch centres enrolling patients with non-rapidly progressive metastatic breast cancer. The study will assess the added value of FES-PET and 89Zr trastuzumab-PET (using tiny quantities of radiolabelled trastuzumab) to predict non-response to targeted treatment with hormone or anti-HER2 therapy before therapy initiation, and of FDG-PET to predict non-response early during drug treatment. “What you want to prove is that it does make sense to get insight into whole-body tumour expression of ER and HER2 to make treatment decisions,” she says.
For de Vries, that study represents only one example of many potential uses for molecular imaging. The technique, she argues, is a perfect tool for understanding how to use targeted therapy. As these therapies, by definition, are designed to hunt down a target, if you want to know the extent to which the target is present in a given patient, all you have to do is circulate a trace amount of the product with a radiolabel attached. Potentially these techniques could also be very helpful to evaluate whether targeted drugs are achieving the desired effect on their target in any given patient. In a number of preclinical studies, de Vries and colleagues have demonstrated the impact of a variety of targeted drugs on the expression of the relevant genes as visualised on PET imaging. They are now conducting clinical trials to visualise the effects of drugs specifically on ER, HER2 and VEGF expression.
“Radiolabelled PET is a perfect tool for understanding how to use targeted therapy”
Molecular imaging can probably even help with identifying the appropriate dose, she says. “For instance, we know from clinical trials, and also from our own work, that if you study the pharmacokinetics of trastuzumab in the blood, it varies considerably from patient to patient. This seems to be related to a large extent to the total tumour volume in a patient, which makes sense: if your antibodies specifically go to tumour lesions, there will be a larger sink for the drug – and therefore more drug required – if you have more tumour on board.” One implication might be that we may be using more trastuzumab than is necessary in adjuvant settings.
Right now de Vries is actively exploring the potential for using fluorescence as an additional cheaper, easier and safer alternative to radioisotopes. The concept is identical to PET scanning, except that the chosen compound is labelled with a fluorescent marker. de Vries says that the advantages are that you don’t need radioactivity, and fluorescence is also better at detecting very small lesions. “You need only a few cells to get the signal. Often for PET scanning you need a lesion to be between 0.5 and 1.0 cm to detect it.” The main problem at the moment is that it is impossible to get a whole-body reading, given the limited penetration of light. “Happily several interesting novel devices are in development that are able to detect fluorescence, for instance during surgery, by endoscopy, with a handheld probe or using diffuse optical tomography to identify fluorescence-labelled lesions in the breast.
de Vries is now keen to join multicentre imaging trials in collaboration with US and European centres. She may feel herself to be something of an alien in this field but the traffic is not all one way. Plenty of imaging specialists are now crossing the border in the other direction to join forces with the clinical cancer community to see how techniques they have spent years developing can function in the real world.
An all-around picture
One of these travellers is the current president of the International Cancer Imaging Society, Anwar Padhani, a radiologist at the Paul Strickland Scanner Centre in London. Padhani shares de Vries’ belief that imaging could offer a vital tool for personalising therapies, but his interest is not so much on imaging molecular targets that may be specific to a cancer phenotype, as building up an all-round picture of how a tumour is sustaining itself and how it is responding to treatment. Learning how to do this effectively could be of enormous benefit to speed up drug development and cut costs as well as making it easier to take informed decisions on the management of individual patients.
Angiogenesis, for instance, is known to be important in delivering the oxygen and nutrients that growing tumours need, and radiologists have developed a technique – dynamic contrast-enhanced (DCE) MRI/CT – that can provide whole-body images of the rate of contrast medium uptake, which is a marker for vascularisation. ‘Before’ and ‘after’ imaging can tell you how effective anti-angiogenic therapies such as bevacizumab and sunitinib are in a given patient. However, further research is needed to see how accurate imaging is at predicting response and patient benefit.
Exciting though this may be, Padhani is looking for something more comprehensive to guide the use of multitargeted therapies. “Just because you alter the blood vessels in a particular tissue doesn’t mean that patients will benefit. You also need to look at what is happening to other processes in the tumour environment. If you kill some cells but make the tumour hypoxic in the process, you can make things even worse, because we know that hypoxic tumours are more resistant.” Getting information on hypoxia requires different types of imaging, such as PET scanning using 18F-misonidazole or 64Cudiacetl- bis (N4-methylthiosemicarbazone). There are also scans that can help show the extent of cell death (such as diffusion-weighted MRI), or levels of proliferation (PET using 18F-fluorothymidine or 11C-choline) or glucose metabolism (FDG-PET).
As many of these processes are linked, it is not always straightforward to interpret the signals. Tumour cells that are starved of oxygen, for instance, tend to respond by switching on more glucose receptors. The FDG-PET scan will tell you where glucose metabolism is upregulated, “but is that because it is more hypoxic or because the tumour phenotype is intrinsically producing more receptors?” Either way, he adds, you know you have an aggressive tumour.
This array of imaging tools offers potential for understanding what a patient needs and how they are responding to selected treatments. But Padhani says there are many challenges to overcome before they can be introduced into clinical practice. These include the issue of how many tests you can do multiple times (cost, logistics and toxicity can be factors here). Then there is the issue of how imaging information is complementary to other biomarkers such as circulating tumour cells, tumour markers, urine biomarkers, immunohistochemistry. “Where does imaging fit in, how does it correlate with these other biomarkers? There are exploratory investigations into this area but they have not progressed far,” says Padhani. He reviewed some of these issues in a paper he co-authored on Multiparametric Imaging of Tumour Response to Therapy, published in Radiology in 2010 (vol 258, pp 348–364).
“There are many challenges to overcome beforethey can be introduced into clinical practice”
There is also a question about proof of clinical benefit. “A lot of this imaging hasn’t yet been correlated with patient outcomes. For FDG-PET we have firm evidence that changes in PET scans actually affect how patients feel and how they survive. This has been shown and in a number of different cancer types, including as a marker of response. But for the vast majority of others it hasn’t been done, and the roadmap of how to do it has not been defined.”
He and his fellow researchers are calling on oncologists to get engaged in this work. “We can’t do it ourselves. We can develop the techniques, but we need active cooperation from the oncologists to be able to take the technique forward, to find its role, what its ‘killer app’ is going to be. The landscape will change and they will need to become much more familiar with imaging as we need to be familiar with what they do. We need to do this together.”
Speeding progrss to the clinic
Efforts to progress the use of imaging in personalised therapies have been concentrated in countries where major research bodies are capable of taking on this task, such as Germany and the UK. Harpal Kumar, chief executive of Cancer Research UK, for instance, recognised in 2008 that “imaging is fast becoming one of the most effective means of detecting cancer early and of determining which treatment works for which patient.” The charity almost quadrupled its funding for this area of work to £50 million (€58 million euros) over five years. A lot of work is also being done in the US, which applies a lighter regulatory hand to the use of new radioactive tracers for investigational procedures.
EU funding for developing imaging biomarkers was boosted in 2006 with the Innovative Medicines Initiative (IMI), a €2 billion EU–industry partnership. Some of this is targeted to:
- create disease-specific European Imaging Networks,
- develop regional centres of excellence, creating disease-specific European centres for the validation of new biomarkers and
- enhance collaboration with patients and regulatory authorities.
The EORTC has already secured funding for a trial investigating the value of diffusion-weighted MRI and PET imaging for proliferation and apoptosis for use as surrogate markers in early clinical trials.
Leading this work is Sigrid Stroobants, head of the Department of Nuclear Imaging at the University of Antwerp, and chair of the EORTC’s Imaging Group, which was established in early 2010. Stroobant’s imaging group scans all new trial proposals submitted to the EORTC to identify opportunities for tacking on an imaging study to the protocol. She says that a lot of observational trials with imaging are needed simply to relate the signals they find to what they see in preclinical studies without interfering in the treatment.
Such imaging add-ons can be very expensive, however – around €500 for an MRI scan, €800 for FDG-PET, and closer to €1000–1500 for other PET tracers, which are not so widely available. There is also a question of capacity. The EORTC imaging group is coordinating with the UK imaging network set up by Cancer Research UK, and between them they cover around 100 centres, but not all of them can do what is required. “Not all clinical centres have the capacity to do these high fancy imaging techniques and you sometimes see a discordance between what we need for imaging and what we need for the clinical department. Sometimes they lack the special sequences we need for diffusion MR or they don’t have access to FLT [18F-fluorothymidine, an alternative PET tracer].”
Stroobants believes that some of the more generic markers that Padhani talks about are good candidates for replacing the traditional RECIST criteria for measuring response in many situations, using FLT- and FDG-PET scanning and probably dynamic contrast-enhanced MRI and diffusion-weighted MRI. She believes that the diffusion-weighted MRI technique may develop to the point where it may start to be used in preference to FDG-PET scans, which are more expensive, involve radioactivity, and are logistically more demanding.
But there is a lot of work to do before this technique can be used in multicentre trials because there is no standardisation yet, and still a lot to learn. “Very simple things that can influence the signals are not known yet. Does the patient need to be fastened in a fixed position or not? What influence does the use of contrast enhancement have? Does it depend on the age of the patient?”
Cross-calibration is required before MRI can be used in multicentre trials, to make sure that differences between images from different centres represent real biological differences and not just different machine settings, and this is one of the work packages from the IMI project. “We hope with the extra funding we received from the EU we will be able to solve that problem, let’s say within one year’s time,” says Stroobants. I’m hoping that within five years we can validate these as biomarkers of response.”
Key to carrying out such multicentre studies will be the imaging platform that EORTC has developed in coordination with Cancer Research UK, which will be used to collect the images centrally and conduct centralised analysis. An imaging ‘warehouse’ has also been established which will link to information on clinical data, tissue, blood and plasma samples stored in biobanks.
The challenge is to find the funding to conduct these trials and to convince clinicians that it is worthwhile taking them on. Stroobants says this can be very hard to do, but that larger multicentre trials are needed. “It is important that we try to incorporate imaging in trials and that we move away from doing single-centre studies and trying to analyse data in our own way. This will not move the field forward. We need to think bigger, multicentre, standardised – the time to play in individual centres is over.”
“We need to think bigger, multicentre, standardised – the time to play in individual centres is over”






Leave a Reply