Stimulating development of more and better paediatric cancer drugs will be key to making progress, particularly in some of the hardest-to-treat childhood cancers. Gilles Vassal, past-President of the European Society for Paediatric Oncology, outlines emerging strategies to make this happen.
This grandround was first presented by Gilles Vassal,
Director of Clinical Research at the Gustave Roussy
Cancer Campus, Villejuif, France, as a live webcast
for the European School of Oncology in collaboration
with the European Society for Paediatric Oncology SIOPE.
It is edited by Susan Mayor. The webcast of this
and other e-sessions can be accessed at e-eso.net.
Cancer is a rare disease in children and young people in Europe, but around 6,000 die each year from the disease. It is the leading cause of death due to disease in children and young people over the age of one year, and is therefore an important public health issue.
Thirty-five thousand young people are newly diagnosed with cancer each year in Europe, of whom 15,000 are children under 15 and 20,000 are young people aged 15–24 years. Eighty percent of them are disease-free at five years across all cancers, with modern treatments and care by a multidisciplinary team.
This means there are currently 300,000 EU citizens who are childhood cancer survivors; about two-thirds of them have long-term side-effects, which are severe in around half.
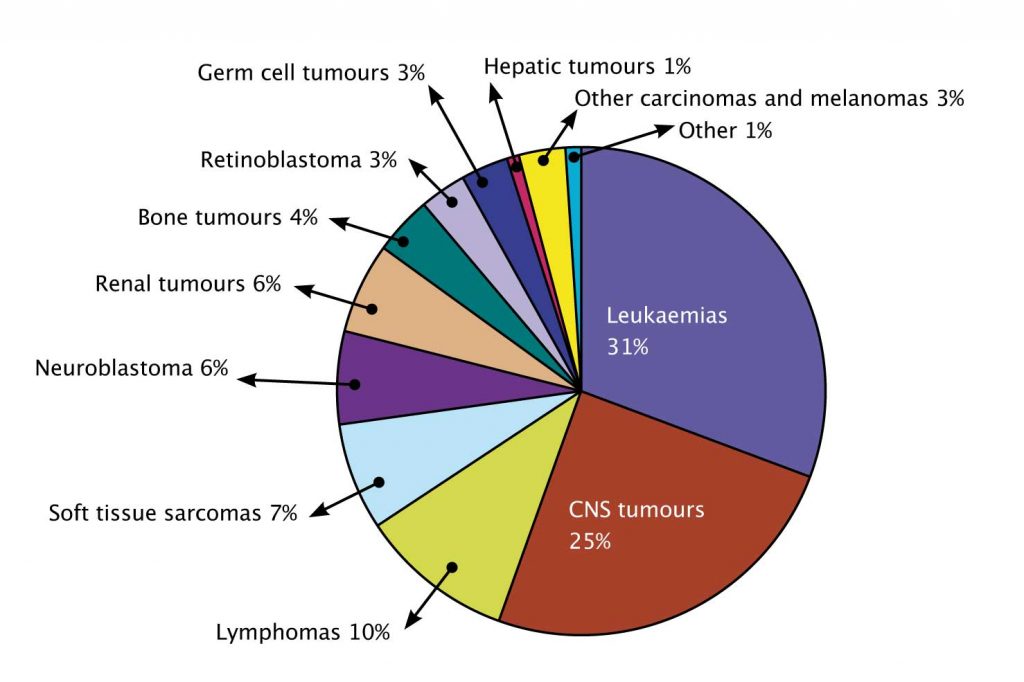
Paediatric malignancies are different from adult cancers. The most frequent cancers in children are leukaemias, followed by central nervous system tumours and lymphomas. More than 60 different cancers occur from newborns to teenagers and beyond, but this number is much greater when considering the different molecular types of each cancer.
For example, there are four different subtypes of medulloblastoma, defined by their molecular biology, which have different prognoses and survival rates and require different treatments. This means that every cancer in a young person is either rare or extremely rare, which impacts on clinical research and the way in which new drugs are developed for patients in this age group.
There are wide variations in survival rates for childhood cancer across Europe, underlining the importance of addressing inequality. Five-year survival is 10–20% lower in some countries than in others, with children in many eastern European countries doing particularly badly compared to others (Lancet Oncol 2014, 15:23–34).
Little progress has been made in hard-to-treat cancers in children over the last 10 years. Medulloblastoma, high-risk sarcoma and high-risk leukaemia, for instance, have shown no improvement in survival, and urgently require new drugs to improve outcomes.
This is why the European Society for Paediatric Oncology (SIOPE) has developed a strategic plan to improve the management of cancers in children and young people over the next 10 years (www.siope.eu/SIOPE_StrategicPlan2015/), with three key goals: to increase cure rates in patients with poor-prognosis malignancies, to improve quality of life in survivors, and to tackle inequalities.
The SIOPE strategic plan has seven objectives:
- Develop innovative therapies for high-risk malignancies.
- Develop and use precision medicine in routine clinical practice, including making use of molecular information from patients’ tumours to drive treatment.
- Improve knowledge of tumour biology to inform treatment and to develop new drugs based on science.
- Provide equal access across Europe for essential medicines and for innovative therapies.
- Address cancers in teenagers and young adults, taking account of their special needs and working with a multidisciplinary team to provide the best quality of care for this age group.
- Improve quality of survivorship.
- Increase understanding of the causes of paediatric cancers, which is necessary to develop new treatments.
Question: Is there currently an issue regarding access to essential medicines in Europe compared to the rest of the world? Are there any countries in Europe that do not have access to essential cancer medicines for children?
Answer: There shortages of older medicines, now produced as generics, in several countries in Europe and other areas. Shortages of these drugs jeopardise the treatment of children with cancer.
Question: With such a high cure rate for many paediatric tumours, is there still room for clinical research in some of these highly curable diseases, such as first-line leukaemias? Or should we focus research on hard-to-treat, relapsed or refractory tumours?
Answer: We definitely need to focus on cancers that are hard to treat at the moment. But treating patients within a prospective protocol is important because it provides the best quality of care and generates knowledge of these diseases, so integration of care and research adds value in paediatric oncology.
Question: What’s the proportion of children compared to adults with cancer that are treated at academic sites, and the proportion treated in private practice in clinical trials?
Answer: Most children (around 95%) and young people with cancer in Europe, are treated in public and university hospitals or cancer centres by paediatric oncologists. We have integrated clinical trials with routine care in paediatric oncology for many years, which means that more than 40% of children are included in clinical trials and more than 80% are treated and prospectively monitored based on standard of care protocols shared by all institutions. This is also a very good context in which to develop new drugs for children with cancer.
Question: What is the role of networking inside and outside Europe, considering that paediatric tumours are so rare? How does collaboration in research on childhood cancers compare with that in adult oncology where there are many more patients and greater opportunities for clinical trials.
Answer: Networking is absolutely crucial in paediatric oncology and has been essential to the significant progress that has been made over recent years. The only way forward to improve care and research is to run trials through co-operative groups at a European or global level rather than at a national level.
Drug development: strategy and organisation
There has been a real explosion in new oncology drugs approved for adults over the last few years, with 70 new drugs approved between 2011 and 2015. While most patients are still not cured of their cancer, and chemotherapy and radiotherapy remain the main modes of therapy, there has been real progress with new drugs based on increased understanding of cancer biology. For example, immunotherapy is completely changing the landscape, with activity across different cancers and offering effective treatment in patients with advanced disease.
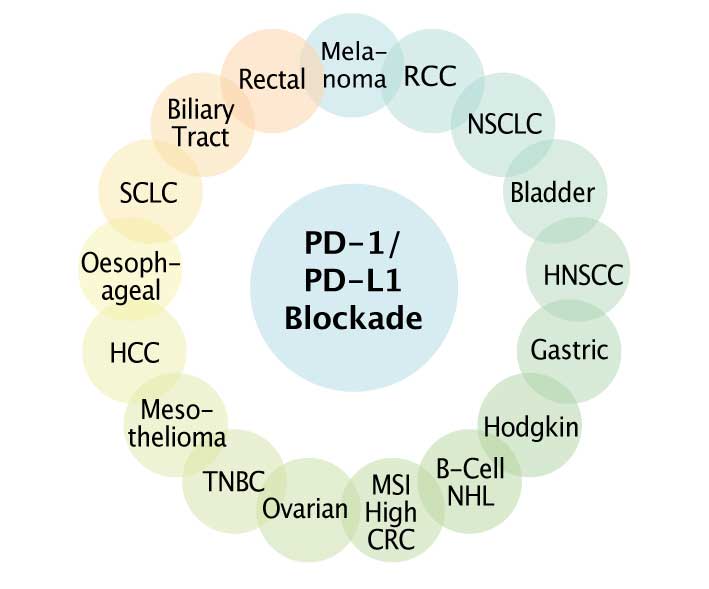
RCC – renal cell carcinoma, – NSCLC non-small- cell lung cancer, HNSCC – head & neck squamous cell cancer, NHL – non-Hodgkin lymphoma, MSI – microsatellite instability, CRC– colorectal cancer, TNBC – triple negative breast cancer, HCC – hepatocellular carcinoma, SCLC – small-cell lung cancer. (Click to enlarge)
PD-1 and PD-L1 blockade is relevant and effective in a wide range of different cancers (see figure). An important question is whether these drugs are also effective in paediatric patients, bearing in mind that these cancers are biologically different from adult tumours, with a much lower rate of mutations (Science 2013, 339:1546).
These biological differences mean that some of the targets for cancer drugs in adults do not exist in children. However, we do have several examples of targets in adult cancers that are also relevant in children, with some shared alterations that can drive drug development. For example B-RAF, which is the target in B-RAF-mutated melanoma, is also mutated in some rare paediatric gliomas. The figure opposite (bottom) shows scans from a patient aged 2.5 years with a peduncular anaplastic BRAFV600 mutated ganglioma, demonstrating a major reduction of the tumour at 19 months after starting treatment with the B-RAF inhibitor vemurafenib.
There are three situations regarding the development of new anticancer drugs for children:
Diseases that are the same in adults and children. Examples include acute lymphoblastic leuk-aemia (ALL), acute myeloid leuk-aemia (AML), chronic myeloid leukaemia (CML), osteosarcoma, and Ewing’s sarcoma. Drug development should occur across all age groups at the same time for these cancers.
Diseases are different but share common targets. The ALK mutation present in some adult lung cancers, for instance, is also present in some paediatric anaplastic large cell lymphoma (ALCL), neuroblastoma and inflammatory myofibroblastic tumours (IMT). The B-RAF mutation present in some adult melanomas is also present in some paediatric ganglioma and histocytosis.
Specific paediatric targets. These include the disialoganglioside GD2 in neuroblastoma, which is an exquisitely specific target that is now being investigated with an agent designed to target it.
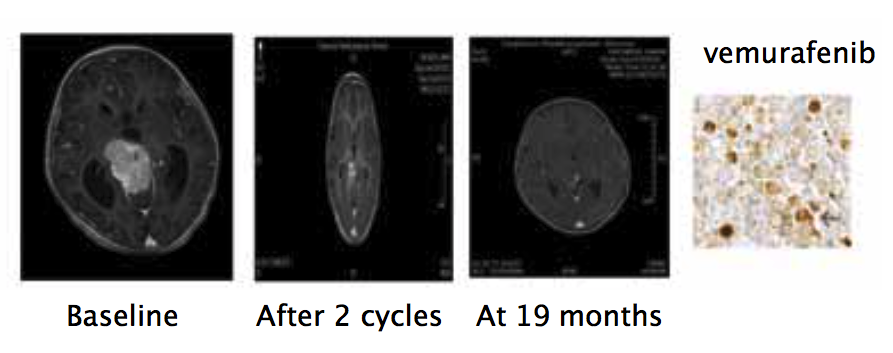
A 2.5-year-old girl with peduncular anaplastic BRAFV600-mutated ganglioma – Targets in adult cancers can also be relevant in children, as in the case of the B-RAF inhibitor vemurafenib, which was approved to treat B-RAF-mutated melanoma, but has also shown efficacy in children with BRAFV600-mutated gliomas
Source: F Bautista et al (2014) Paediatric Blood Cancer 61:1101–03, John Wiley and Sons (click to enlarge)
Understanding cancer biology is essential for developing new drugs, and molecular information will increasingly be used for guiding treatment choice. Europe is ready to run a biology-driven new drug development strategy with well-organised networks.
The Innovative Therapies for Children with Cancer European Consortium (www.itcc-consortium.org) has been set up to carry out phase I and early phase II trials for drug registration. It is a consortium of 52 institutions in 13 countries across Europe and Israel, working in collaboration with other networks in specific cancers.
These networks include the IBFM Study Group in lymphoma and the SIOPE Brain Tumour Group. In addition, there is co-operation with groups in the US and globally, providing the scale to develop drugs for rare cancers.
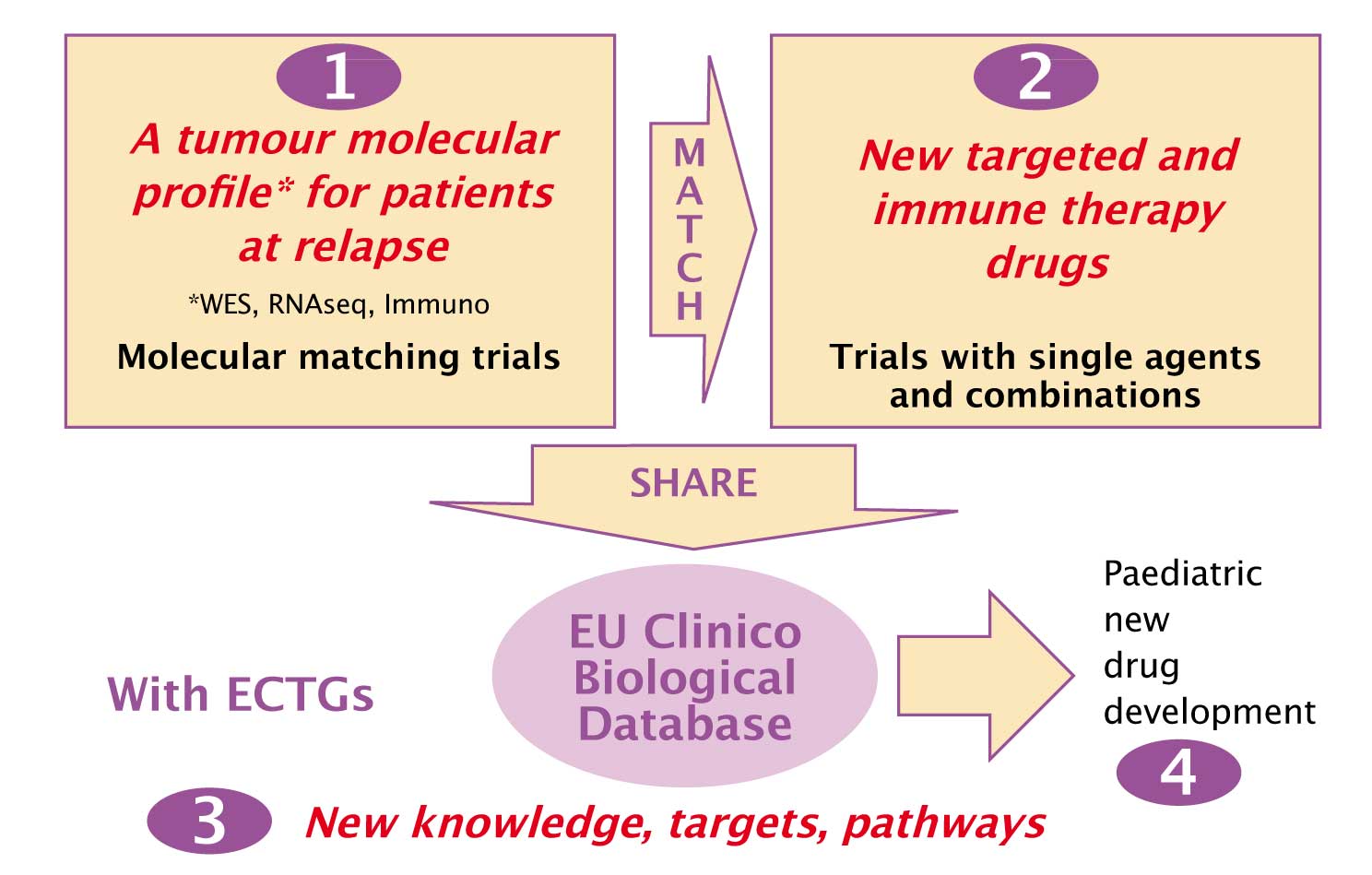
The strategy (see figure) is firstly to perform a tumour molecular profiling for each individual patient at relapse. Then clinical and biological data are collected in a large European database to generate new knowledge, targets and pathways.
Molecular profiling information in paediatric tumours is now being collected and matched to drug targets in four major studies in Europe as part of the ITCC Precision Cancer Medicine programme: INFORM (Germany); MAPPYACTS (France, Spain, Denmark and Italy), iTHER (Netherlands) and S-PED (UK). The number of phase I and phase II trials being conducted by the ITCC is increasing, and includes the MATRIX trial, providing rapid access to atezolizumab and cobimetinib, and the ESMART trial, which was launched in August 2016 with seven treatment arms based on five drugs provided by several companies for children with molecular alterations in relapsed cancers.
Question: How would you compare the collaboration between academic consortia and industry in paediatric oncology with that in adult cancers?
Answer: It’s new for industry to work with paediatric oncologists and for paediatric oncologists to work with industry. This is a real opportunity to work out how best to collaborate together, along with parents and regulatory bodies, to develop new drugs in a way that may be different to the process in adult cancers. Co-operation is essential to do our best for the development of innovative treatments in childhood cancers, where patient populations are often small.
Question: The number of actionable targets is relatively limited in childhood cancers. How do you envisage giving access to innovative drugs where there is no molecular target yet identified, so these children can benefit?
Answer: Only 15–20% of children have alterations in tumours that are targets for drugs that we can identify at the moment. However, we have set up studies to provide access to treatments for children, whether their tumour has a drug target or not. This is because molecular alterations with targeted drugs, such as B-RAF or ALK, are very rare, so we are generating molecular information in all patients considered appropriate for treatment with innovative compounds.
Question: Development of single agents is necessary to study pharma–cokinetics, efficacy and safety, especially in children. However, cancer requires multimodal treatment – radiation, surgery and combination chemotherapy. Combinations have been studied less in children than in adults. How can the development of combinations be accelerated?
Answer: We definitely need to accelerate the development of combi-nation treatments. In the ESMART trial, six of the seven treatment arms are combinations including new drugs. We are moving to new types of trial, for example with the first cycle being single-agent therapy and the second cycle being combination therapy including new drugs. We need to carry out more trials with combination therapies and, in addition, move rapidly from trials in relapsed disease to first-line treatment trials. For example, we are currently running a trial in children with diffuse intrinsic pontine gliomas, introducing new drugs with radiotherapy at diagnosis.
Improving the regulatory environment
The current regulatory landscape for paediatric medicines was set out in EU regulations published in 2007. At that time, developments in new drugs for childhood cancers were based on drugs that were already available, with little input from pharmaceutical companies. Current approaches to develop innovative treatments require effective collaboration, working together more effectively to develop new drugs in paediatric oncology.
The 2007 regulations aimed to ensure high-quality research into the development of medicines for children and to ensure, over time, that the majority of medicines used by children are specifically authorised for such use. However, 50% of the drugs we currently use to treat children with cancer are used off-label. The regulations approved drugs for children at the time of submission for marketing authorisation in adults, based on the obligatory requirement for a paediatric investigation plan, which can be waived or deferred in certain cases. If completed, this allows for a six-month extension of market exclusivity, whatever the results.
This was a ‘stick and carrot’ system that clearly worked, illustrated by the increasing number of drugs available to the ITCC through paediatric investigation plans. A single drug (imatinib), when it was set up in 2003, increased to 12 drugs in 2013. However, only two new targeted drugs have been authorised for use in cancer in children over the last 10 years.
There are three key issues that need to improve in the regulatory environment. The first of these concerns unjustified class waivers, such as for crizotinib, which in Europe secured a waiver of the requirement to develop a paediatric investigation plan because the disease it treats (ALK-positive non-small-cell lung cancer) does not occur in children – even though a phase I trial in children showed responses in relapsed anaplastic large-cell lymphoma, inflam-matory myofibroblastic tumours and neuroblastoma.
The European regulation ignored the fact that the drug could be active in malignancies other than lung cancer, including paediatric cancers.
The second regulatory issue that needs revisiting is where paediatric investigation plans prove unfeasible. The vemurafenib paediatric investigation plan, EMA/193393/2011, for example, was unable to recruit.
Thirdly, delays in starting paediatric drug developments also need to be addressed.
To improve the regulation of drugs in children, a working group has made a number of suggestions (EJC 2016):
- Paediatric development should be based on a drug’s mechanism of action instead of the adult indication.
- Drugs should be prioritised for evaluation in children according to the mechanism of action, needs and feasibility.
- Decisions should be made through a multi-stakeholder forum, using stronger biological and preclinical data.
- New incentives and rewards should be offered for developing new drugs for paediatric use, and measures introduced to reduce the time before starting paediatric drug development.
Conclusion
In summary, we need to improve the regulations for developing new drugs to treat children with cancer. In addition, we need a change in mind set, moving innovation in paediatric medicines from an issue of regulatory compliance to one of research and development, working collaboratively and facilitating referral of children to centres conducting relevant clinical trials. In addition, we need to invest in the development of new oncology drugs specifically for children.





Leave a Reply