Patients who are receiving adequate nutrition have a better prognosis, respond better to chemotherapy and can tolerate higher doses of anticancer treatments. It is therefore important for oncologists to assess and manage malnutrition.
 Malnutrition, which is easily identified during clinical examination by weight loss and hypophagia, is an independent negative prognostic factor for cancer patients. Nutritional health can be considered based on a person’s protein status. This is very important because there is no store of protein in the body, yet each protein has a specific function, for example as an enzyme, antibody, or contracting muscle protein or transport protein. The severity of malnutrition is often related to the degree of the protein depletion.
Malnutrition, which is easily identified during clinical examination by weight loss and hypophagia, is an independent negative prognostic factor for cancer patients. Nutritional health can be considered based on a person’s protein status. This is very important because there is no store of protein in the body, yet each protein has a specific function, for example as an enzyme, antibody, or contracting muscle protein or transport protein. The severity of malnutrition is often related to the degree of the protein depletion.
Nutritional health can be defined as having 100% of body protein (see figure overleaf). Depending on the duration of starvation or the cause of malnutrition, protein depletion leads to loss of organ function. This starts with decreased muscle mass (skeletal, cardiac and smooth muscle), followed by decreased visceral proteins, including albumin, transferrin and transport protein. Further protein malnutrition results in impaired immune response, which is compromised with the decrease of lymphocytes and synthesis of antibodies and acute phase proteins. This can be seen in a surgical patient, with impaired wound healing. The next step in protein depletion is impairment of organ function – gut, liver and heart. Finally, further protein depletion leads to a poor adaptation to any minimal biologic stress, which can prove to be fatal. Nitrogen death was defined thirty years ago as when 30% of body nitrogen has been lost. This depletion is incompatible with survival.
So-called secondary malnutrition (the type commonly associated with a serious infectious or neoplastic disease) leads to protein depletion, and differs from pure starvation, such as in anorexia nervosa, where visceral proteins are maintained and remain stable until weight loss is extreme. In cancer or sepsis, where there is an inflammatory status, the decrease of visceral proteins is common.
Several studies have identified malnutrition as an independent negative prognostic factor for survival in patients with a variety of malignancies. We have very extensive evidence to show that malnutrition plays a major role in predicting poor prognosis, based on measuring weight loss,[1] low bioelectric phase angle[2–10] or depletion of body protein or fat with sophisticated laboratory methods.[11,12] On clinical grounds, the Prognostic Nutritional Index[13,14] and the Glasgow Prognostic Score15 are very effective in identifying patients with a poor prognosis.
Malnutrition not only compromises survival, but also has a major adverse effect on the quality of life. Studies have shown that malnourished cancer patients have:
- higher rates of hospital readmissions and longer hospital stays[16,17]
- increased symptom distress[18]
- reduced quality of life, based on usual questionnaire for cancer patients[19–24]
- reduced muscle strength and functional status[8]
All of these studies have identified malnutrition as an independent factor that adversely affects the quality of life.
Importantly for oncologists, malnutrition increases chemotherapy toxicity. This has been demonstrated for weight loss and hypoalbuminemia[25] and low total body nitrogen as a predictor of neutropenia.[26] It has also been demonstrated for sarcopenia as a significant predictor of toxicity, based on CT scan.[27] It is also true for patients with a body mass index (BMI) lower than 25kg/m².[28] All of these factors have been found to be associated with poor adherence to chemotherapy, and high toxicity.
Malnourished cancer patients also have poorer responses to chemo‑ therapy, both in terms of the percentage of patients responding to chemotherapy and the duration of response to treatment.[29,30] The fact that malnourished patients have a poor prognosis, are more likely to have poor responses to chemotherapy, and have increased toxicity means that it is important for oncologists to assess and manage malnutrition.
 Question: We have so much data, going back more than 40 years, so why are people still trying starvation diets to kill their cancer and why is the awareness of the negative impact of malnutrition still so low among oncologists?
Question: We have so much data, going back more than 40 years, so why are people still trying starvation diets to kill their cancer and why is the awareness of the negative impact of malnutrition still so low among oncologists?
Answer: When patients die because of cancer there is often a combination of cachectic status due to the inflammatory reaction that we know is a major contributor to the weight loss, metabolic derangement and poor nutrient intake. For oncologists and other clinicians it is not clear whether a cancer patient has died because of tumour progression or starvation. Some patients with a slowly progressing tumour or a tumour not involving vital organs could survive for some months, but they die sooner because they do not eat enough. The problem is related to the fact that it is difficult to separate the morbidity and mortality that is due to the simple deficiency of macronutrients from the alteration of metabolism that is due to inflammation, which is a major cause of cachexia. So many oncologists have a nihilistic approach and do not try to feed cancer patients in an optimal way. In contrast, I suggest that these patients should be supported in the best possible way with nutrition.
How can we identify cancer patients at nutritional risk?
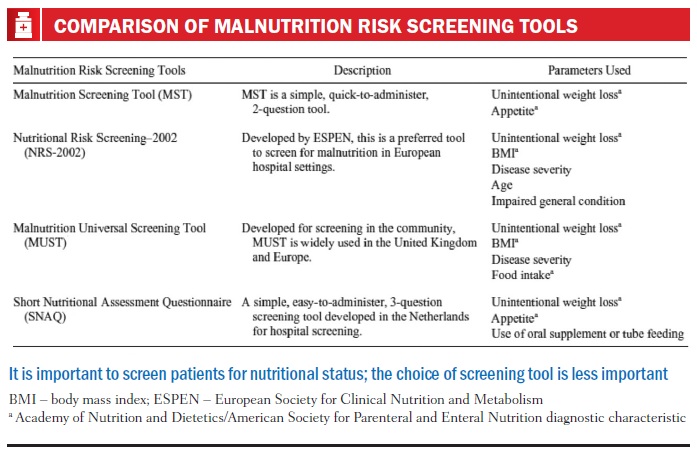
There are several nutritional screening tools, but the most important and most commonly used in hospitals are shown in the table above. The Malnutrition Screening Tool (MST) relies mainly on unintentional weight loss and appetite, so it is very simple to use this score if a hospital has limited resources. The Nutritional Risk Screening includes more parameters: unintentional weight loss, BMI, severity of disease, age, and impaired general condition, with scores ranging from 0 to 7. An important point about this screening tool, which is commonly used in Europe, is that it was developed to identify not only malnourished patients but also those who may improve with nutritional support.
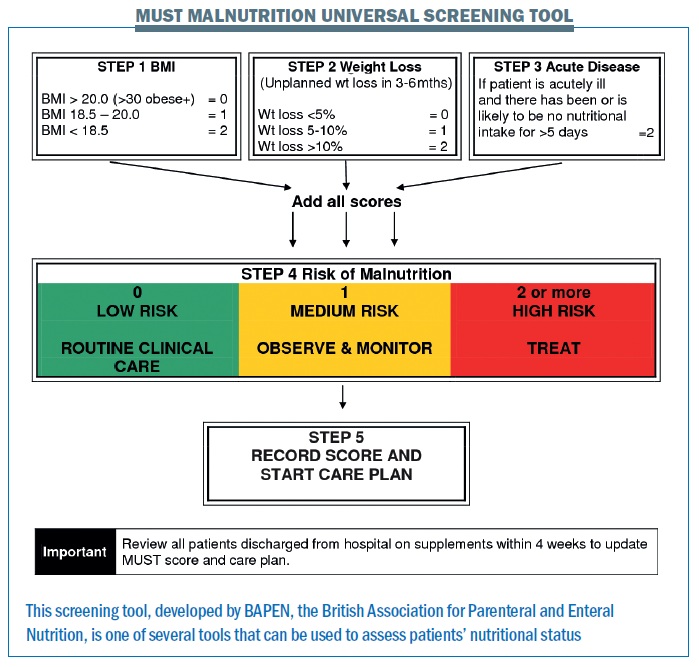
The Malnutrition Universal Screening Tool (MUST) is widely used in the UK and Europe, and includes unintentional weight loss, BMI, severity of disease and food intake. The Short Nutritional Assessment Questionnaire (SNAQ) asks questions about unintentional weight loss, appetite and use of oral supplements or tube feeding. A very interesting and comprehensive review on screening tools by Marian van Bokhorst[31] found there is no perfect screening tool, and none of the tools are better than the others, but concluded that the important thing is to use a tool to assess patients from a nutritional point of view.
Question: Which malnutrition risk screening tool would you recommend?
Answer: It depends on the situation. I used the Nutritional Risk Screening 2002 tool (NRS-2000) for my studies, and we published at least two studies demonstrating that nutritional risk is correlated to the type and stage of a patient’s tumour. In routine clinical practice, if I realise that a patient is anorexic because they say they have no appetite, relatives report that the patient is not eating and the patient has lost weight, this information is enough to consider nutritional support. However, if you want to stratify for trials of nutritional support, I would recommend the NRS-2002, though this is not because it has been demonstrated to be better than the others. Ideally in routine practice a patient’s chart should include a space to report their nutritional risk.
 Ways to provide nutritional support to cancer patients
Ways to provide nutritional support to cancer patients
The approach to nutritional support depends on the availability of a working/accessible gastrointestinal (GI) tract. Very simply, we can consider nonsurgical cancer patients identified as malnourished at nutritional risk in three broad groups:
- patients with the whole GI tract working
- patients with the upper GI tract inaccessible
- patients with the whole GI tract inaccessible or not working.
In patients with the whole GI tract working, I think the first approach is oral nutritional intervention with supplements, which are better if enriched with omega-3 or leucine, with or without dietetic counselling and megestrol. However, sometimes we may also consider supplementary intravenous nutrition. In patients who already have a central line it is sometimes easier to give nutritional supplementation by vein than forcing oral intake or using a tube.
What are the effects of oral supplementation?
Dietary counselling alone does not ameliorate quality of life, but diet-ary counselling plus nutritional supplements improves weight more than dietary counselling alone or usual care.[32–34] Dietary counselling plus nutritional supplements improves quality of life, according to two studies.[35–37] However, oral nutritional intervention has no effect on cancer mortality.
An experimental diet high in protein and enriched with 10% free leucine was found to significantly improve the muscle protein fractional synthesis rate compared to a standard diet.[38] A study giving amino acids rapidly in high quantity (40 g), given as a bolus, increased the mixed muscle fractional synthesis rate in cancer patients undergoing intense chemotherapy.[39] These studies show that giving amino acids as a bolus or an enriched leucine diet can improve muscle synthesis in cancer patients, despite chemotherapy or inflammatory status.
There is some controversy over supplements enriched with omega-3 fatty acids, according to four systematic reviews and two meta-analyses.[40–45] One meta-analysis concluded that omega-3 supplementation increased lean body mass in cancer patients, while the other found no improvement. Recent non-randomised clinical trials have shown that omega-3 fatty acids increased lean body mass in patients with head and neck cancer[46] and increased the muscle mass, body weight and response to chemotherapy in patients with lung cancer.[47]
Recent randomised controlled trials, not included in the previous meta-analyses, demonstrated improved quality of life in patients with lung cancer,[48] as well as reduced leukopenia in patients on neoadjuvant chemotherapy for oesophageal cancer,[49] and reduced weight loss and higher remission rates in leukaemic patients receiving omega-3 fatty acids.[50]
How can we optimise the use of oral nutritional supplements? An excellent systematic review[51] found greater adherence to higher-energy-density supplements (91% with 2 kcal/ml). Adherence was probably better with liquid oral nutritional supplements. This sort of energy supplement should be in addition to food, with clinical benefits when the intake was in the range of 300–600 kcal/day for more than five weeks.
Dietary energy density was positively associated with energy balance. Survival was positively associated with energy balance while systemic inflammation had a negative association. The review recommended using omega-3 fatty acids and/or leucine-enriched oral nutritional supplements. When amino acids are used, they should be given as a bolus.
 Patients with inaccessible upper GI tract
Patients with inaccessible upper GI tract
Options for patients with an inaccessible GI tract are tube feeding using either a nasogastric tube or percutaneous endoscopic gastrostomy, where the tube feeds directly into the patient’s stomach, passing through their abdominal wall.
There is a lot of experience in patients with head and neck cancer, during radiation with or without chemotherapy, and many non-randomised trials report better weight maintenance and quality of life, as well as better adherence to therapy and fewer hospital admissions, compared with oral feeding.
A randomised clinical trial comparing percutaneous endoscopic gastrostomy with use of a nasogastric tube found that percutaneous endoscopic gastrostomy was associated with better weight maintenance and a longer duration of enteral nutrition[52,53] as well as a similar[52] or better[53] quality of life. The results are quite limited so we cannot recommend percutaneous endoscopic gastrostomy over nasogastric feeding.
Patients whose whole GI tract is inaccessible or not working
You are obliged to use parenteral (intravenous) nutrition in patients whose GI tract is inaccessible or not working. There is little scientific experience and very few randomised trials, but the approach is very practical and well accepted by those patients who already have a central venous catheter and may not be able to differentiate between therapy and nutritional support. This may be important from a psychological point of view, as the patients do not realise that they are so compromised that they require nutritional support to survive. Small-volume high-density emulsions can cover a large part of the patient’s energy requirement, so they can be used easily in home environments.
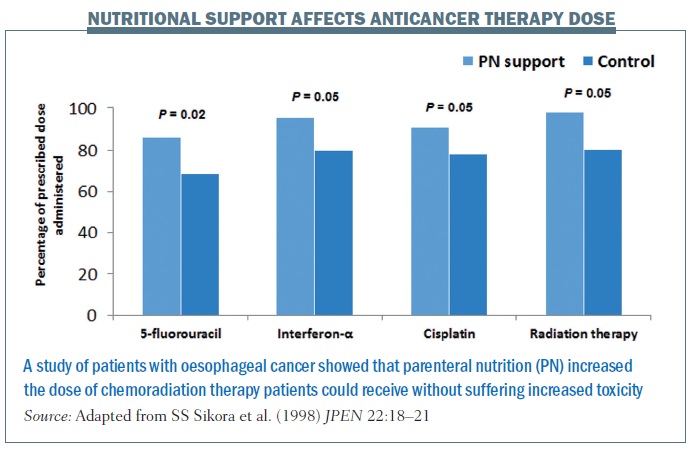
Supplemental parenteral nutrition can be useful in patients whose GI tract is only partially obstructed or who are partially aphagic (have a reduced ability to swallow), because delivery via a vein may be more comfortable for them and is easier than putting a tube in the stomach or forcing oral nutrition. A randomised study in patients with cancer of the oesophagus showed those fed by parenteral nutrition were able to receive higher doses of chemoradiation therapy (see figure below) without increased toxicity, compared to controls.[54]
A recent Chinese study identified malnourished cancer patients using a nutritional screening tool and treated them with enteral or parenteral nutrition. In comparison with patients who did not receive any nutritional treatment, those who received enteral or parenteral nutrition had a significantly reduced risk of developing adverse events. The authors concluded that undernutrition and nutritional risk are common problems that impact on outcomes of hospitalised cancer patients.[55]
There are few studies on the use of supplemental parenteral nutrition at home, but one study showed giving supplemental parenteral nutrition intravenously in cachectic patients who were not totally aphagic when their oral intake dropped to 21–24 kcal/kg/day was associated with an increase in energy balance, longer survival and improved maximum exercise capacity.[56] A further study demonstrated an increase in lean body mass in cancer patients receiving supplemental parenteral nutrition.[57]
We conducted a study in 414 incurable cancer patients who were cachectic and almost aphagic using parenteral nutrition at home. Results showed a six-month survival of 28% and a three-month survival of 57%.[58] In contrast, according to the literature, without nutritional support these patients would have survived less than three months.
We were also able to identify some simple biochemical/clinical prognostic factors that predict a higher rate of three-or six-month survival in patients on home total parenteral nutrition. A randomised trial cannot be carried out for ethical reasons, but comparison with evidence in the literature suggests that survival can be prolonged with parenteral nutrition at home. Home parenteral nutrition may therefore prolong survival in selected incurable cancer patients who are cachectic and aphagic, usually with malignant obstruction, and who do not have jaundice or major liver, renal or respiratory insufficiency.
According to the guidelines of both the American Society for Parenteral and Enteral Nutrition and the European Society for Clinical Nutrition and Metabolism, the routine use of enteral or parenteral supplementation during chemotherapy is not recommended. However, if patients are malnourished or facing more than a week of starvation, then oral nutritional supplements and/or enteral nutritional support should be considered (grade B recommendation, with supporting evidence in the literature). If this is not feasible, then parenteral nutrition is recommended.
If patients develop GI toxicity from chemotherapy or radiation therapy, short-term parenteral nutrition may be better tolerated (and more efficient) than enteral nutrition to restore intestinal function, prevent nutritional deterioration and allow full adherence with therapy (general consensus statement).
References
1. J Bachmann et al. (2008) Cachexia worsens prognosis in patients with resectable pancreatic cancer. J Gastrointest Surg 12:1193–20
2. MB Viana MB et al. (1994) Malnutrition as a prognostic factor in lymphoblastic leukaemia: a multivariate analysis. Arch Dis Child 71:304–310
3. MP Davis et al. (2009) Bioelectrical impedance phase angle changes during hydration and prognosis in advanced cancer. Am J Hosp Palliat Care 26:180–187
4. D Gupta et al. (2004) Bioelectrical impedance phase angle as a prognostic indicator in advanced pancreatic cancer. Br J Nutr 92:957–962
5. D Gupta et al. (2004) Bioelectrical impedance phase angle in clinical practice: implications for prognosis in advanced colorectal cancer. Am J Clin Nutr 80:1634–38
6. D Gupta et al. (2008) Bioelectrical impedance phase angle as a prognostic indicator in breast cancer. BMC Cancer 8:249
7. D Gupta et al. (2009) Bioelectrical impedance phase angle in clinical practice: implications for prognosis in stage IIIB and IV non-small cell lung cancer. BMC Cancer 9:37
8. K Norman et al. (2010) Cutoff percentiles of bioelectrical phase angle predict functionality, quality of life, and mortality in patients with cancer. Am J Clin Nutr 92:612–619
9. SI Paiva et al. (2009) Standardized phase angle from bioelectrical impedance analysis as prognostic factor for survival in patients with cancer. Support Care Cancer 19:187–192
10. L Santarpia et al. (2009) Prognostic significance of bioelectrical impedance phase angle in advanced cancer: preliminary observations. Nutrition 25:930–931
11. MG van Vledder et al. (2012) Body composition and outcome in patients undergoing resection of colorectal liver metastases. Br J Surg. 99:550–557
12. RA Murphy, MS Wilke MS, M Perrine et al. (2010) Loss of adipose tissue and plasma phospholipids: relationship to survival in advanced cancer patients. Clin Nutr. 29:482-487
13. T Nozoe et al. (2002) Correlation of preoperative nutritional condition with postoperative complications in surgical treatment for oesophageal carcinoma. Eur J Surg Oncol 28:396–400
14. T Nozoe et al. (2010) Prognostic Nutritional Index: a tool to predict the biological aggressiveness of gastric carcinoma. Surg Today 40:440–443
15. ABC Crumley et al. (2006) Evaluation of an inflammation-based prognostic score in patients with inoperable gastro-oesophageal cancer. Br J Cancer 94:437–441
16. MI Correia and DL Waitzberg DL. (2003) The impact of malnutrition on morbidity, mortality, length of hospital stay and costs evaluated through a multivariate model analysis. Clin Nutr 22:235–239
17. M Pressoir et al. (2010) Prevalence, risk factors and clinical implications of malnutrition in French Comprehensive Cancer Centres. Br J Cancer 102:966–971
18. L Sarna et al. (1994) Weight change and lung cancer: relationship with symptom distress, functional status, and smoking. Res Nurs Health 17:371–379
19. HJN Andreyev et al. (1998) Why do patients with weight loss have a worse outcome when undergoing chemotherapy for gastrointestinal malignancies? Eur J Cancer 34:503–509
20. P O’Gorman, DC McMillan and CS McArdle. (1998) Impact of weight loss, appetite, and the inflammatory response on quality of life in gastrointestinal cancer patients. Nutr Cancer 32:76-80
21. G Ollenschlager et al. (1992) Nutritional behaviour and quality of life during oncological polychemotherapy: results of a prospective study on the efficacy of oral nutrition therapy in patients with acute leukaemia. Eur J Clin Invest 22:5–23
22. L Ovesen et al. (1993) Effect of dietary counseling on food intake, body weight, response rate, survival, and quality of life in cancer patients undergoing chemotherapy: a prospective, randomized study. JCO 11:2043
23. CR Persson et al. (2002) A randomized study of nutritional support in patients with colorectal and gastric cancer. Nutr Cancer 42:48–58
24. A Nourissat et al. (2008) Relationship between nutritional status and quality of life in patients with cancer. Eur J Cancer 44:1238–42
25. O Arrieta et al. (2010) Association of nutritional status and serum albumin levels with development of toxicity in patients with advanced non-small cell lung cancer treated with paclitaxel-cisplatin chemotherapy: a prospective study. BMC Cancer 21:10:50
26. A Aslani et al. (2000) The predictive value of body protein for chemotherapy-induced toxicity. Cancer 88:796–803
27. CM Prado et al. (2008) Prevalence and clinical implications of sarcopenic obesity in patients with solid tumours of the respiratory and gastrointestinal tracts: a population-based study. Lancet Oncol 9:629–635
28. S Antoun et al. (2010) Low body mass index and sarcopenia associated with dose-limiting toxicity of sorafenib in patients with renal cell carcinoma. Ann Oncol 21:1594–98
29. F Di Fiore et al. (2007) Baseline nutritional status is predictive of response to treatment and survival in patients treated by definitive chemoradiotherapy for a locally advanced esophageal cancer. Am J Gastroenterol 102:2557–63
30. E Mitry (2004) Predictive factors of survival in patients with advanced colorectal cancer: an individual data analysis of 602 patients included in irinotecan phase III trials. Ann Oncol 15:1013–17
31. MAE van Bokhorst-de van der Schueren et al. Nutrition screening tools: Does one size fit all? A systematic review of screening tools for the hospital setting. Clin Nutr 33:39–58
32. TR Halfandarson et al. (2008) Does dietary counseling improve quality of life in cancer patients? A systematic review and meta-analysis. J Support Oncol 6:234–237.
33. C Baldwin and CE Weekes. (2012) Dietary counselling with or without oral nutritional supplements in the management of malnourished patients: a systematic review and meta-analysis of randomised controlled trials. J Hum Nutr Diet 2012 25:411–426
34. C Baldwin, A Spiro, R Ahern et al. (2012) Oral nutritional interventions in malnourished patients with cancer: a systematic review and meta-analysis. J Natl Cancer Inst 104:371–385
35. EA Isenring, S Capra and JD Bauer. (2004) Nutrition intervention is beneficial in oncology outpatients receiving radiotherapy to the gastrointestinal or head and neck area. Br J Cancer 91:447–452
36. P Ravasco et al. (2005a) Impact of nutrition on outcome: a prospective randomized controlled trial in patients with head and neck cancer undergoing radiotherapy. Head Neck 27:659–668
37. P Ravasco et al. (2005b) Dietary counseling improves patient outcomes: a prospective, randomized, controlled trial in colorectal cancer patients undergoing radiotherapy. JCO 23:1431–38
38. NE Deutz et al. (2011) Muscle protein synthesis in cancer patients can be stimulated with a specially formulated medical food. Clin Nutr 30:759–68
39. EL Dillon et al. (2007) Amino acid metabolism and inflammatory burden in ovarian cancer patients undergoing intense oncological therapy. Clin Nutr 26:736–743
40. P Mazzotta and CM Jeney. (2009) Anorexia-cachexia syndrome: a systematic review of the role of dietary polyunsaturated fatty acids in the management of symptoms, survival, and quality of life. J Pain Symptom Manage 37:1069–77
41. BS van der Meij, MA van Bokhorst-de van der Schueren and JA Langius. (2011) N-3 PUFAs in cancer, surgery, and critical care: a systematic review on clinical effects, incorporation, and washout of oral or enteral compared with parenteral supplementation. Am J Clin Nutr 94:1248-65
42. C Persson, B Glimelius, J Rönnelid. (2005) Impact of fish oil and melatonin on cachexia in patients with advanced gastrointestinal cancer: a randomized pilot study. Nutrition 21:170–178
43. A Ries, P Trottenberg, F Elsner et al. (2012) A systematic review on the role of fish oil for the treatment of cachexia in advanced cancer: An EPCRC cachexia guidelines project. Palliat Med 26:294–304
44. R Colomer, JM Moreno-Nogueira, PP García-Luna et al. (2007) N-3 fatty acids, cancer and cachexia: a systematic review of the literature. Br J Nutr 97:823–831
45. A Dewey, C Baughan, T Dean. (2007) Eicosapentaenoic acid (EPA), an omega-3-fatty acid from fish oils) for the treatment of cancer cachexia (Cochrane Review) In: The Cochrane Library, Issue 2, Oxford: Update Software
46. HG Weed et al. (2011) Lean body mass gain in patients with head and neck squamous cell cancer treated perioperatively with a protein- and energy-dense nutritional supplement containing eicosapentaenoic acid. Head Neck. 33:1027–33
47. RA Murphy et al. (2011) Supplementation with fish oil increases first-line chemotherapy efficacy in patients with advanced nonsmall cell lung cancer. Cancer Feb 15. doi: 10.1002/cncr.25933
48. BS Van der Meij et al. (2008) A double blind randomized controlled trial on oral nutritional supplementation of omega-3 fatty acids in non-small cell lung carcinoma. Clin Nutr Suppl 3:111
49. H Miyata, et al. Randomized study of clinical effect of enteral nutrition support during neoadjuvant chemotherapy on chemotherapy-related toxicity in patients with esophageal cancer. Clin Nutr 31: 330–336 50330–336
50. I Bayram et al. (2009) The use of a protein and energy dense eicosapentaenoic acid containing supplement for malignancy-related weight loss in children. Pediatr Blood Cancer 52:571–574
51. GP Hubbard et al. (2012) A systematic review of compliance to oral nutritional supplements. Clin Nutr 31:293–312
52. J Corry et al. (2008) Randomized study of percutaneous endoscopic gastrostomy versus nasogastric tubes for enteral feeding in head and neck cancer patients treated with (chemo)radiation. J Med Imaging Radiat Oncol. 52:503–510
53. E Silander. (2012) Impact of prophylactic percutaneous endoscopic gastrostomy on malnutrition and quality of life in patients with head and neck cancer: a randomized study. Head Neck 34:1–9
54. SS Sikora et al. (1998) Role of nutrition support during induction chemoradiation therapy in esophageal cancer. JPEN 22:18–21)
55. H Pan et al. (2013) The impact of nutritional status, nutritional risk, and nutritional treatment on clinical outcome of 2248 hospitalized cancer patients: a multi-center, prospective cohort study in Chinese teaching hospitals. Nutr Cancer 65:62–70
56. K Lundholm et al. (2004) Palliative nutritional intervention in addition to cyclooxygenase and erythropoietin treatment for patients with malignant disease: effects on survival, metabolism and function. Cancer 100:1967–77
57. U Pelzer et al. (2010) Parenteral nutrition support for patients with pancreatic cancer. Results of a phase II study. BMC Cancer 10:86
58. F Bozzetti et al. (2014) The prognosis of incurable cachectic cancer patients on home parenteral nutrition: a multi-centre observational study with prospective follow-up of 414 patients. Ann Oncol 25:487–493





Leave a Reply