Thrombosis is the second most common preventable cause of death in patients with cancer, so oncologists need to know how to identify who is at risk, and strategies for prevention and treatment. This overview presents the evidence and raises alerts about the use of oral anticoagulants in the cancer setting.
The bidirectional relationship between cancer and thrombosis has been known about for nearly 150 years, since Armand Trousseau first identified the link. However, despite being the second most common and preventable cause of death in outpatients with cancer, until recently cancer-associated thrombosis (CAT) has been a largely undiagnosed and undertreated condition. Cancer patients have a four- to seven-fold increased risk of venous thrombo-embolism (VTE) compared to the general population, with the highest risk in the first few months after cancer diagnosis. The incidence is high and increasing, with 20–30% of all first VTEs being cancer related.
It is important to be clear what we’re talking about. Studies are still hampered by the lack of standardisation of detection and reporting of VTE. The International Society on Thrombosis and Haemostasis (ISTH) sub-committee on malignancy defines acute cancer-associated thrombosis as “diagnosis of the index DVT [deep-vein thrombosis] or PE [pulmonary embolism] was made within the past 1 month” (JTH 2013; 11:1760–65). DVT needs to be fully defined as to whether it is symptomatic, proximal, which limb is affected, and which blood vessel. Similarly, pulmonary embolism needs to be defined by whether it involves a segmental or more proximal pulmonary artery, with some counting sub-segmental arteries as well. It is also essential to define what we mean by other related terms, including recurrence, extension of the thrombosis and incidental thrombosis.
Clinical presentation
Cancer-associated thrombosis can be quite debilitating for patients. Most thromboses are asymptomatic, but a cancer registry with more than 10,000–15,000 patients shows that most patients with DVT present with extremity oedema (80%), pain (75%) and erythema (26%) (Haematologica 2008; 93:273–278). Patients with pulmonary embolism present with shortness of breath (85%) and chest pain (40%). Catheter-associated VTE has similar signs and symptoms, with the addition of catheter dysfunction (JCO 2003; 21:3665–75).
The adverse consequences of cancer-associated thrombosis include: increased risk of early death; compromised quality of life; more frequent hospital visits; need for anti-coagulation, which can cause bleeding complications; increased healthcare costs; increased risk of post-phlebitic syndrome and greatly increased risk of recurrent thrombosis. In addition, patients may have to interrupt potentially life-saving cancer treatment.
Should we screen patients presenting with VTE for cancer? Acute VTE can be the first manifestation of an occult cancer. There have been many small studies looking at using extensive screening, baseline screening or no screening at all. We know that patients with unprovoked VTE are at higher risk of having cancer, but no studies have found screening to be cost effective or to affect patient survival. At the moment in our practice, we do an abdominal ultrasound in patients deemed to be at risk of a malignancy.
Risk factors for VTE in cancer patients
One risk factor for thrombosis in cancer patients is the tumour type. There is a higher incidence in patients with ovarian, stomach and pancreatic cancers, particularly with advanced disease; patients with breast or prostate cancers or melanoma are among those with the lowest risk.
Patient-related risk factors include age (although there have been conflicting reports on this), immobility, previous VTE, and comorbidities. Women have a higher risk of VTE, and some patients have prothrombotic gene mutations. Treatment-related factors also influence VTE risk. Surgeons are generally good at giving prophylactic anticoagulation, sometimes for extended periods, for cancer-related surgery. Some chemotherapies cause increased risk of VTE. Hormone therapies, for example tamoxifen, and some of the new anti-angiogenic agents such as VEGF inhibitors and immunomodulatory agents, are associated with increased risk of arterial thrombosis and a slightly higher risk of venous thrombosis. Lenalidomide and thalidomide in combination with dexamethasone for patients with multiple myeloma also increases VTE risk. More details on risk factors can be found in A Young et al. in Nature Reviews Clinical Oncology (9:437–449).
Although we have known the risk factors for some time, predicting the risk of VTE in individual cancer patients is difficult.
Treatment of VTE in cancer patients
It is essential to involve the patient and their family and carers in treatment decisions. The CLOT trial reported in 2003 that low-molecular-weight heparin (LMWH; dalteparin was used in the trial) is superior to warfarin or other vitamin K antagonists (NEJM 2003; 349:146–153), but many patients with cancer-associated thrombosis are still treated with warfarin throughout the world. Alternatives include unfractionated heparin (UFH) for patients with renal impairment, and fondaparinux for patients with heparin-induced thrombocytopenia (HIT). The meta-analysis done by Ellie Akl and the Cochrane database shows that this is the treatment we should be giving, and yet many centres do not (Cochrane Reviews 2011; 15:CD006650).
What about treatment of recurrent venous thromboembolism? Cancer patients have a three-fold greater risk of recurrent VTE than the general population (Blood 2002; 100:3473–88).
Treating recurrent thrombosis is a real problem in the clinic. Patients on subtherapeutic anticoagulation with warfarin or LMWH can be switched to full-dose LMWH (see figure). Patients on therapeutic anticoagulation with warfarin should shift to LWMH, and those already on LMWH should have the dose increased by 20–25%, according to expert opinion from the ISTH malignancy subcommittee.
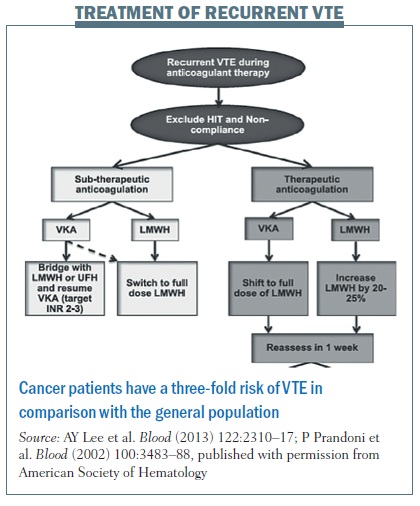 Patients should be reassessed after one week. Those showing symptomatic improvement can continue with the increased dose of LMWH. Measure anti-Xa levels in patients without symptomatic improvement to see if you can increase the dose of LMWH.
Patients should be reassessed after one week. Those showing symptomatic improvement can continue with the increased dose of LMWH. Measure anti-Xa levels in patients without symptomatic improvement to see if you can increase the dose of LMWH.
How long do we treat the patients with a thrombosis? Decisions are based on the balance of bleeding versus thrombosis. Other considerations include the status of the patient’s cancer – whether they’ve got early or advanced disease – type of treatment, impact on quality of life and patient preference. The updated ASCO guidelines for VTE management in cancer patients (2013) recommend considering 12 months anticoagulation when treating symptomatic VTE in patients with advanced or metastatic disease. However, if the increased risk remains, you could consider treatment for the rest of the patient’s lifespan. There are no trials clarifying the duration of anticoagulation, but two UK studies have just started looking at duration of treatment in cancer-associated thrombosis. – ALICAT (www.controlled-trials.com/ISRCTN37913976) and select-d (www.controlled-trials.com/ISRCTN86712308).
Prophylaxis of VTE in cancer patients
Are the rates of VTE high enough to warrant prophylaxis? Studies show varying rates of thromboembolism in cancer patients, with control rates of around 15% before 2000 and 5% or less in recent studies. The SAVE-ONCO study published last year compared prophylaxis with the ultra-LMWH semuloparin with placebo (NEJM 2012; 366:601–609). Results showed a significantly lower rate of VTE with semuloparin (1.2% vs 3.4% with placebo; P=0.001), but the FDA decided not to promote this ultra-LMWH, as the results did not clarify which cancer patients would most benefit, given the side-effect of bleeding. Major bleeding occurred at similar rates with placebo (1.1%) and semuloparin (1.2%). We need more trials on VTE prophylaxis, with large numbers of patients at high risk of VTE.
Current guidelines (ESMO, ACCP, NCCN and ASCO) recommend against routine thromboprophylaxis in outpatients with cancer. In patients who have additional risk factors and who are at low risk of bleeding, they suggest prophylactic doses of LMWH or unfractionated heparin. Additional risk factors are: previous VTE, immobilisation, hormone therapy, angiogenesis inhibitors, and treatment with thalidomide and lenalidomide with dexamethasone.
Question: For patients at high risk, are there any anticancer drugs, other than lenalidomide, that are high risk for thrombosis?
Answer: Thalidomide derivatives are high risk. And if we are using erythropoietin-stimulating agents, we give prophylaxis. Apart from these two classes of drugs, I think risk should be assessed on an individual basis.
Question: Regarding the cost, would using low-dose aspirin be beneficial?
Answer: We don’t use low-dose aspirin at all for prophylaxis and treatment of venous thromboembolism. It is cheap but has most benefit for arterial thrombosis. However, clinicians do recommend its use in other parts of the world, especially in Asia.
Question: Regarding anti-Xa levels, when you decide to increase the level of LMWH for patients with recurrent VTE, is there any target for the anti-Xa levels you should be trying to reach?
Answer: There’s great debate in the UK at the moment, each laboratory has a different assay, and you have to go with your own laboratory to determine peak levels. Getting laboratories to do the same assays would be good.
Risk prediction tools
How do we risk assess the individual patient? Based on consensus, the most recent ASCO guidelines recommend that patients with cancer (outpatients, as well as inpatients) be assessed for their thrombosis risk at the time of starting chemotherapy and periodically after this (JCO 2013; 31:2189–2204). Risk should be assessed using a validated risk assessment tool. In the UK, all hospitalised patients – not just cancer patients – undergo a simple, government-mandated, risk assessment for VTE. So we already risk assess all inpatients but not outpatients.
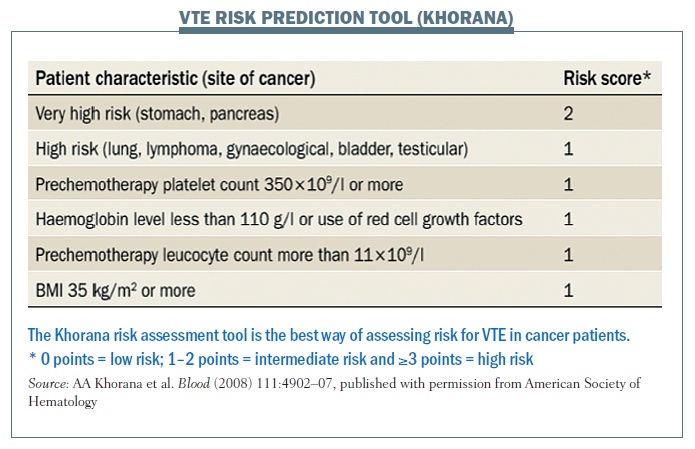
Alok Khorana was the first to develop a risk prediction tool a few years ago, stemming from a neutropenic sepsis study that he was doing that confirmed the risk factors we have previously covered. Patients are scored for their risk factors (see figure below), with a Khorana score of 0 being low risk, a score of 1–2 points being intermediate risk and a score of 3 or more considered higher risk, when you would consider giving prophylaxis. This tool has been validated by studies in two countries – the Austrian Cancer And Thrombosis Studies (CATS) and SENDO (South European New Drugs Office) phase I studies in Italy. The Austrian team added two more risk factors – p-selectin and d-dimer (Blood 2010; 116:5377–82), but these have not been validated as yet. We do not currently use the Khorana risk tool in the UK, but only the simple Department of Health generic tool; however, we should, certainly in our centre, as so far it is the best tool we’ve got.
There is another clinical prediction tool for risk stratification for recurrent VTE in patients with cancer (Circulation 2012; 126:448–454) based on two observational studies. High-risk predictors – the sex of the patient, the primary tumour site, the stage, and prior VTE – all score +1. Low-risk predictors score negative points – breast cancer scores -1, and stage 1 disease scores -2 points. Scores of <0 are low risk and >1 are high risk, when you would consider anticoagulation. This needs to be validated by other teams. We are starting to risk stratify for the individual patient.
 Catheter-related thrombosis
Catheter-related thrombosis
Catheter-related thrombosis is not yet clearly defined: is it a blood clot in the lumina of the catheter, round about the catheter, or a mural thrombus that has gone right across the vein? We have to define what we are talking about, because rates of thrombosis in catheter studies vary widely. In a meta-analysis of warfarin versus control in catheter-related studies we published in 2009 (Lancet 373: 567–574), the confidence intervals crossed the line of unity and the difference was not significant. Although early studies showed that warfarin was better, these were tiny studies, and larger studies showed that low-dose warfarin (1 mg) does not reduce the rates of catheter-related thrombosis, with similar findings for LMWH (JCO 2005; 23:4063–69). So we do not recommend – and the ASCO guidelines say this as well – prophylaxis for catheter-related thrombosis, certainly not with warfarin and only with LMWH if there are other risk factors.
 Survival benefit
Survival benefit
Since the 1970s we’ve been looking to see if there is there any survival benefit – do anti-coagulants designed to have an anti-coagulant effect and not an anti-neoplastic effect have any impact on patient survival? A meta-analysis of all relevant studies, published in 2012 – most of them small and therefore underpowered for survival – found no survival benefit of anti-coagulation (NEJM 2012; 366:661–662). Some sub-studies and some analyses done post hoc showed that specific populations of patients may benefit, but these require further definition. The biological rationale for a heparin effect is emerging.
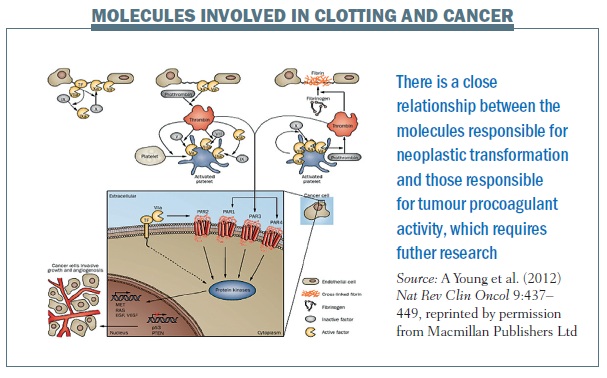
As well as the clinical predictors and the risk factors for VTE, there are also laboratory biomarkers. These are useful in identifying high-risk patients that may benefit from prophylaxis. These biomarkers encompass: factors that activate at the clotting system, such as d-dimer and p-selectin; factors indicating increase in the inflammatory potential around the milieu of the tumour, such as the leucocyte and platelet count; and initiation of the clotting cascade, which can be tested for by measuring tissue factor expressing microparticles. A recent study showed that TNF-alpha is a candidate gene contributing to VTE pathogenesis in gastrointestinal cancer patients (Ann Oncol 2013; 24: 2571–75), so we’re now looking at gene studies to see what contributes to VTE pathogenesis.
The figure below illustrates the close relationship between the molecules responsible for neoplastic transformation and tumour procoagulant activity, which we need to research further. Thrombin generated by the coagulation cascade activates cell surface receptors such as PAR1. The extracellular domain of tissue factor binds to factor VIIa and starts off the clotting pathway. The intracellular domain changes start the signal transduction that modifies and modulates cancerous cells through many pathways including the protein kinases and the MAP kinase pathway.
 How to manage tricky cases of cancer-associated thrombosis
How to manage tricky cases of cancer-associated thrombosis
Expert opinion from the ISTH malignancy subcommittee can help with the management of cancer-associated thrombosis in clinical practice (JTH 2013; 11:1760–65). However, there are no studies to help with this, as yet.
Symptomatic recurrent VTE
Recommend: If patient is on vitamin K antagonists, switch to LMWHs.
Suggest: If on therapeutic LMWHs, use a higher dose (25%), and assess in 5–7 days.
Suggest: If no symptomatic improvement, use peak anti-Xa level to estimate next dose escalation.
Thrombocytopenia
Recommend: Full therapeutic dose anticoagulation, if the platelet count is ≥50×109/l.
Recommend: For acute cancer-associated thrombosis and platelet count <50×109/l, full therapeutic dose anticoagulation with platelet transfusion.
Bleeding
Recommend: Careful and thorough assessment of each bleed. Supportive care with transfusion and surgical intervention to stop bleeding where possible.
Stop anticoagulation.
Suggest: IVC (interior vena cava) retrievable filter in patients with acute or subacute cancer-associated thrombosis with major bleeding.
Novel oral anticoagulants (NOACs)
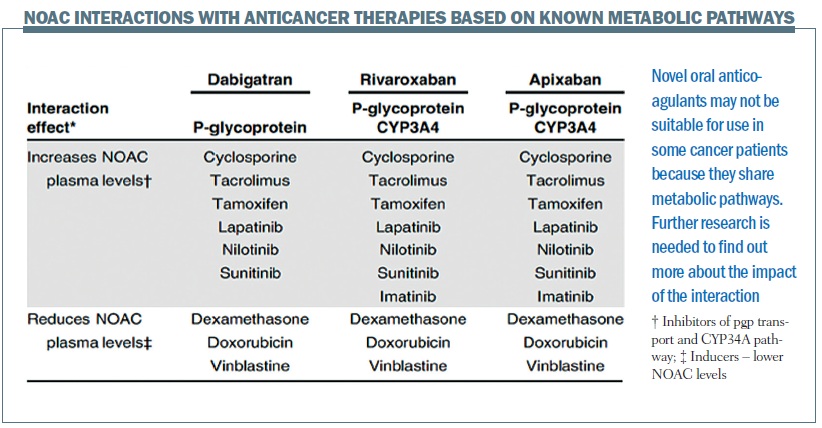 NOACs don’t come without their concerns. They are metabolised through the P-glycoprotein pathways and also the cytochrome P450 3A4 (CYP34A) pathway, so we try to avoid their use with anticancer agents metabolised in the same way (see figure above). Some anticancer therapies, for example sunitinib and imatinib, interact with the P-glycoprotein and the CYP3A4 pathways, but we do not know if that translates into any clinical effect, so studies need to be carried out. We are carrying out a pilot study (select-d) comparing the NOAC rivaroxaban with dalteparin in patients with active cancer and VTE at first randomisation, stratifying by risk factors. Patients who are positive for residual vein thrombosis (RVT) at six months (patients with DVT and all PE patients) will then continue treatment, randomised to rivaroxaban or placebo, while those with no evidence of RVT will stop (JTH 2012; 10:807–814).
NOACs don’t come without their concerns. They are metabolised through the P-glycoprotein pathways and also the cytochrome P450 3A4 (CYP34A) pathway, so we try to avoid their use with anticancer agents metabolised in the same way (see figure above). Some anticancer therapies, for example sunitinib and imatinib, interact with the P-glycoprotein and the CYP3A4 pathways, but we do not know if that translates into any clinical effect, so studies need to be carried out. We are carrying out a pilot study (select-d) comparing the NOAC rivaroxaban with dalteparin in patients with active cancer and VTE at first randomisation, stratifying by risk factors. Patients who are positive for residual vein thrombosis (RVT) at six months (patients with DVT and all PE patients) will then continue treatment, randomised to rivaroxaban or placebo, while those with no evidence of RVT will stop (JTH 2012; 10:807–814).






Leave a Reply