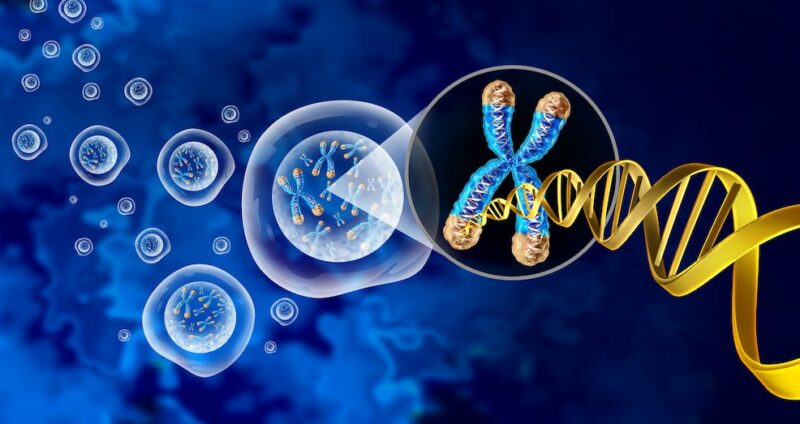
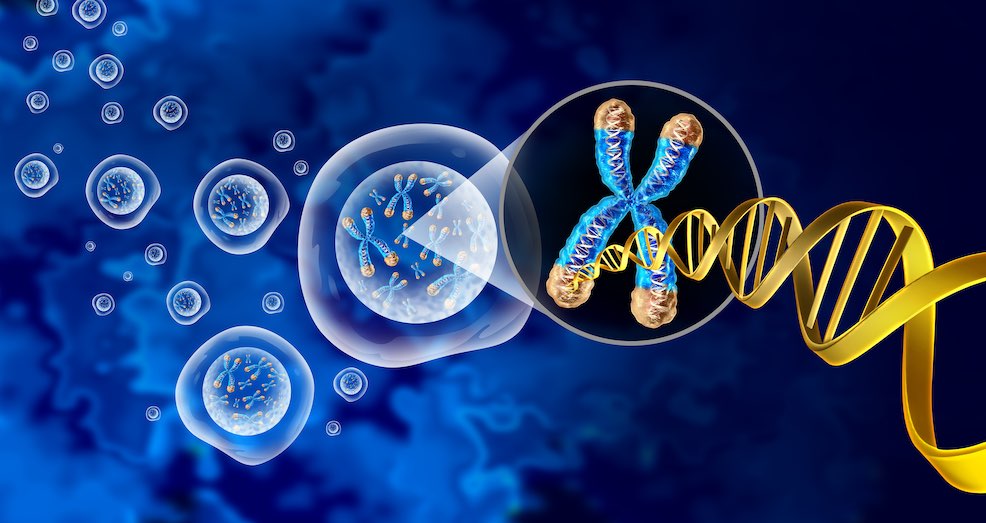
Shortening of telomeres, as occurs naturally with ageing, works through a pathway that involves mitochondria-activated immune responses to ultimately eliminate precancerous cells. The US study, published in Nature (8 February 2022), suggests that telomere shortening has evolved as a tumour-suppressive mechanism, and when this complex pathway becomes dysfunctional, cells evade destruction and become cancerous.
“These findings highlight a synergy between critically short telomeres, mitochondria and innate immunity that has evolved to prevent age-associated cancer initiation in humans,” write the authors, led by Jan Karlseder and Gerald Shadel, from the Salk Institute for Biological Studies, La Jolla, California.
The study, Karlseder tells Cancerworld, also emphasizes the importance of caution with use of medical drug treatments to lengthen telomeres. “You just don’t want to be messing with the length of telomeres, because the outcome is likely to be cancer,” warns Karlseder. Issues, he adds, are unlikely to arise from exercise, which a recent systematic review published in 2022 in Medicina found results in increased telomere length, as the actual increases here were minimal.
Elizabeth Blackburn together with Carol Greider and Jack Szostak won the 2009 Nobel Prize in Physiology or Medicine for the discovery of how chromosomes are protected by telomeres and the enzyme telomerase. In an earlier study published in Nature in 2019, the Karlseder lab examined the effects of short telomeres in a state called ‘crisis’, where unprotected chromosomes become dysfunctional and fuse to each other. Cells in crisis, they went on to show, are removed by a process called autophagy, in which the body rids itself of damaged cells.
In the current study, Karlseder teamed up with Shadel, whose lab studies the role mitochondria play in human disease, and went on to explore further the activation mechanism of autophagy-dependent cell-death during crisis when telomeres are extremely short.
Starting from the hypothesis that short telomeres cause cell death, the team reasoned that if they suppressed the regulators of cell death, cells would survive. Utilizing a human fibroblast tissue culture (where they understood dynamics for cell growth and crisis), they conducted a genetic screen where they systemically suppressed approximately 19,000 genes using commercially available CRISPR libraries. Eventually the innate immune sensor Z-DNA binding protein 1 (ZBP1) was identified as a regulator of crisis.
Next, by marking the ZBP1 protein with immunofluorescence, they demonstrated that it located to the outer mitochondrial membrane.
At the level of the mitochondrial membrane the team went on to show that ZBP1 activates the innate immune adaptor protein mitochondrial antiviral-signalling protein (MAVS), which in turn amplifies the inflammatory process that ultimately triggers autophagy. They accomplished this by demonstrating that disruption of MAVS prevented inflammation and autophagy activation. As a proof of principle, they demonstrated that, when MAVS was disrupted, ZBP1 was still able to gravitate to the mitochondria, but that no inflammation or cell death occurred. This indicates that MAVS represents a critical intermediate regulator of the process.
“Taken together we showed that, when telomeres become critically short, they are recognised by the sensor ZBP1, then in response ZBP1 localises to mitochondria where it leads to an amplification of an inflammation signal that in turn activates autophagy, resulting in cell death,” explains Karlseder. “This pathway has evolved to remove cells with critically short telomeres from the population, which would otherwise go on to gather an unstable genome and potentially become cancerous.”
While the team acknowledge that they are only just starting to unravel the complex process, they believe that it may help develop novel cancer therapeutics. “Ultimately, we believe that if we could mimic short telomeres in cancer cells that have become resistant to therapy, we could activate pathways to specifically kill such cells,” says Karlseder.
Next, the team plan to explore whether disrupting autophagy in a mouse model with shortened telomeres results in cancer formation. “We need to demonstrate that the model works in an in vivo system not just in vitro in tissue culture cells,” says Karlseder.