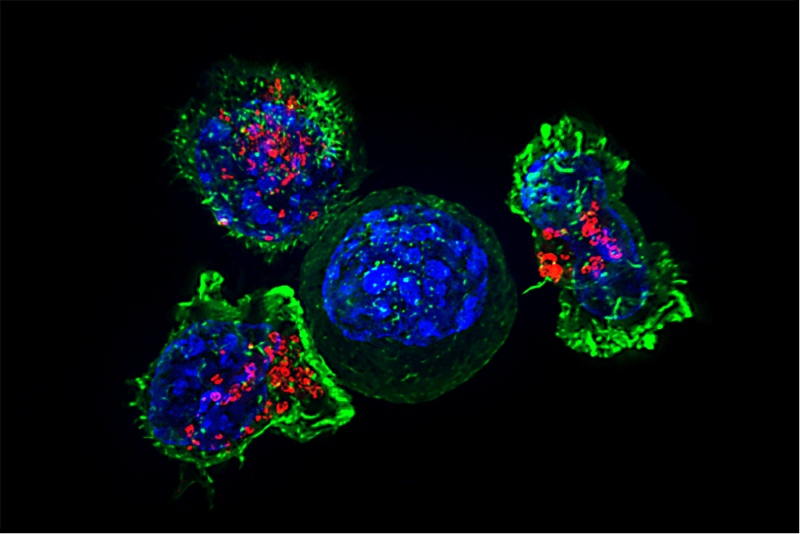
A study published in PNAS (Chen IX, et al. PNAS 2020) of a new mouse model has enabled the identification of distinct gene signatures that distinguish responders from non-responders to immune checkpoint blockade (ICB).
“Our findings provide a general approach for investigating mechanisms of resistance to ICB and identifying predictive biomarkers of response,” write the authors Rakesh Jain, Meromit Singer, and colleagues from Harvard Medical School, Boston, MA.
Although ICB has been approved by the FDA in more than 20 indications, only a subset of patients have been found to derive any clinical benefit. For example, more than 75% of patients with breast cancer who received either anti-PD-1 or anti-PD-L1 treatment did not show any objective response. Therefore, there is intense interest in identifying and developing predictive biomarkers of ICB response.
In the current study, the investigators report on a new bilateral mouse tumour implantation model permitting both resection of the tumour as well as evaluation of treatment response. This is the first time this model has been used in breast cancer “to assess the entire immune compartment within the tumour microenvironment in a resection and response-type model”, explains Rakesh Jain. For the study, bilateral breast cancer tumours were implanted into the mammary fat pads of mice, and the animals were treated with an anti-PD1 antibody or a control IgG antibody. A tumour on one side was removed to allow for evaluation of the tumour microenvironment, while the tumour on the other side was kept in situ to monitor treatment response and enable the mouse to be identified as a responder or a non-responder.
Results showed that responder mice had higher levels of CD8+ T-cell infiltration in the tumour microenvironment and that transcriptomic analysis of CD8+ T cells revealed distinct gene signatures that distinguished responders from non-responders. Furthermore, these gene signatures for responders and non-responders correlated significantly with responder and non-responder gene signatures derived from melanoma patients who were treated with ICB, as well as overall survival in breast cancer patients treated with ICB.
“Based on these analyses and our observation that patient survival rates significantly associated with our generated responder and non-responder signatures, we envision that future studies could construct a classifier that utilizes distinct attributes of these human-adjusted signatures to assist with patient diagnostic and treatment decisions,” write the authors.
• • • A second study, reported in Nature Communications (de Streel G et al. Nat. Comms 2020) demonstrated how selective blockade of the TGF-β1 receptor by regulatory T cells (Tregs) induced regression of mouse tumours otherwise resistant to anti-PD-1 immunotherapy. The blockade was achieved by utilizing antibodies against GARP: TGF- β1complexes. The team, led by Sophie Lucas, from the Université Catholique de Louvain, additionally showed that GARP-expressing Tregs are present in a sizeable subset of human melanoma samples. “Taken together, our results support the clinical evaluation of blocking anti-GARP:TGF-β1 mAbs, administered alone or in combination with other therapeutic strategies, to treat patients with cancer resistant to currently available immunotherapies,” write the authors. A phase I trial (NCT03821935), they add, was recently initiated to test such antibodies in the clinic.
Image credit: Alex Ritter, Jennifer Lippincott Schwartz and Gillian Griffiths, National Institutes of Health. Republished under a Creative Commons Licence.