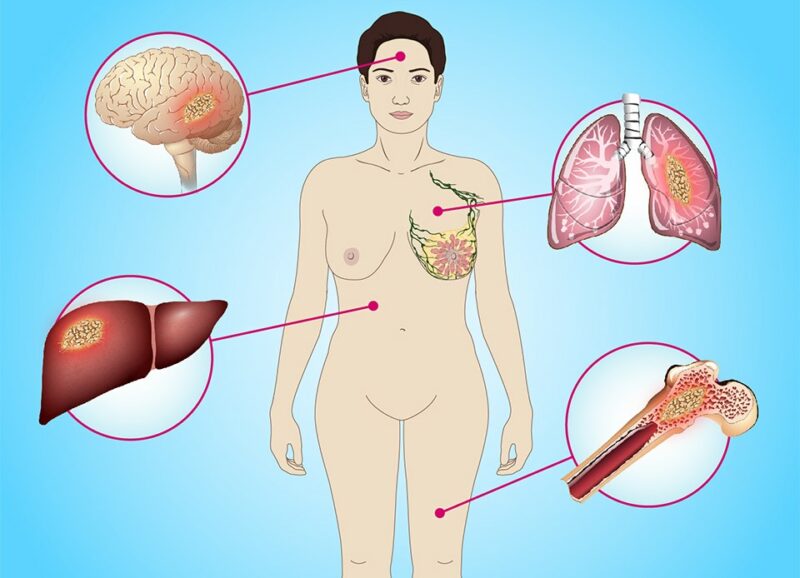
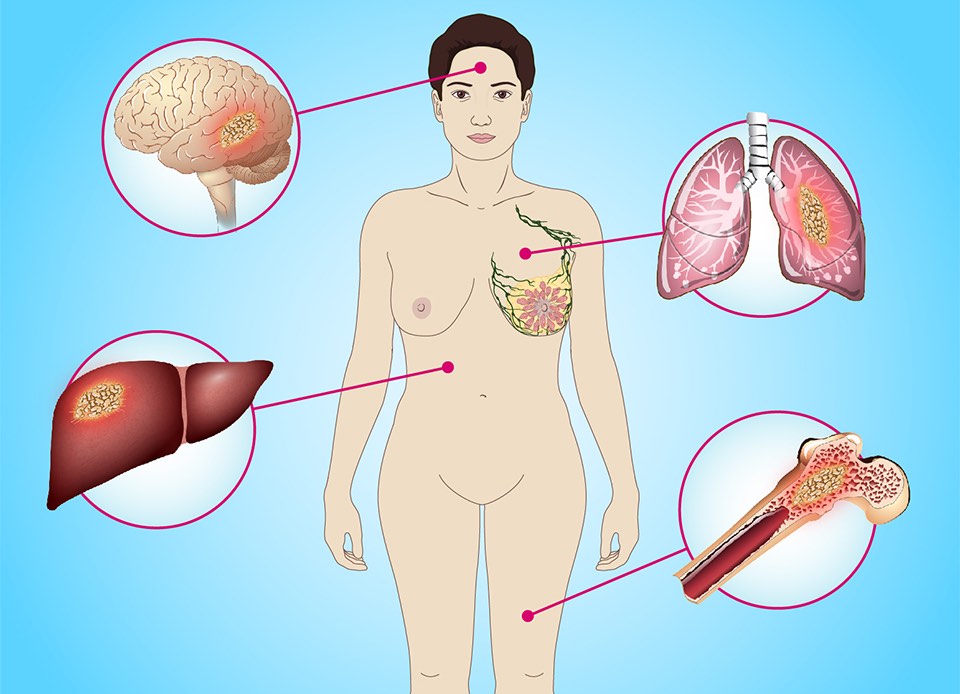
Around one third of breast cancer patients showed molecular subtype shifts (especially involving luminal cancers) between their primary and metastatic tumours. The study, published in Nature Cancer, on December 30th, also revealed the presence of fewer immune cells in triple-negative (TNBC)/ basal-like metastases compared to primary tumour microenvironments. Together, the findings suggest immune therapies may not be appropriate for all patients with TNBC metastatic breast cancers.
“We have found that the same patient can have a completely different molecular subtype of breast cancer in the metastatic setting from the primary setting, which changes how their tumour should be treated,” says Susana Garcia-Recio, co-lead author, from University of North Carolina, Chapel Hill. “The lesson from our study is that, whenever possible, metastatic breast cancer patients should be having their metastatic tumours re-analysed to plan future lines of therapy, possibly warranting a different therapeutic strategy.”
Metastatic breast cancer is one of the leading causes of cancer-related mortality among women in the Western world. So far, most research efforts have focused on molecular analysis of the primary tumour to dissect genotypes of the disease. However, accumulating evidence supports a molecular evolution of breast cancer during its life cycle, with metastatic lesions acquiring new molecular aberrations.
The complimentary AURORA US and AURORA EU projects, launched in 2014, were established to help the research community identify those at risk of metastasis before it occurs, or much earlier, and find new, more effective therapies or improve response to existing ones. AURORA US aims to characterise the molecular underpinnings driving tumour evolution from the primary tumour in the breast to distant, life-threatening disease; while AURORA EU is focused on the evolving molecular landscape during tumour evolution with a particular emphasis on how tumour evolution affects response to therapies.
For the current retrospective study, the AURORA US team identified 55 women from nine US institutions, for whom fresh-frozen metastatic specimens were available that could be linked to primary tumours already banked as fresh-frozen or formalin-fixed paraffin-embedded samples. Of the 55 women, who had received a median of three lines of systemic therapy and had a median overall survival from diagnosis of 4.5 years, 19 had more than one metastasis analysed.
In the study, tumour DNA and RNA were isolated from each specimen and used in four different assays: DNA exome and low-pass whole-genome sequencing (WGS; tumour and normal); whole transcriptome RNA sequencing (using rRNA depletion); and DNA methylation microarrays. “Using the four platforms allowed us to have a more comprehensive view and look at a range of factors including differences in gene expression patterns and the tumour microenvironments,” explains Garcia-Recio.
Altogether, the team analysed 51 primary cancers and 102 metastases, which were obtained from multiple sites, with the most common being liver (n=28), lung (n=13), lymph nodes (n=12), brain (n=11), and other (n=16). In total, 88 of the 153 specimens had all four assays successfully performed, and 141 had three of the four completed.
Key findings from the study included:
- Expression subtype changes were observed in around 30% of samples and were coincident with DNA clonality shifts, especially involving HER2.
- Down-regulation of oestrogen receptor (ER)- mediated cell–cell adhesions through DNA methylation mechanisms was observed in metastases.
- The immune system was found to be diminished in the metastatic samples. In TNBC/basal metastases, a decrease in B and T cells was observed.
- In the metastatic setting, the gene human leukocyte antigen-A (HLA-A), known to be used by the immune system to recognise malignant cells, showed lower expression in comparison to the primary setting. Furthermore, DNA methylation patterns and focal deletions (the loss of part of the HLA-A gene), were found on this gene. This lack of expression resulted in fewer immune cells present in the tumour microenvironment being able to recognise and attack metastatic tumours.
Three examples of clonal evolution were highlighted in the paper, including participant ARF3, who was diagnosed with an ER+/PR+/HER2– LumA breast cancer, and was treated with chemotherapy and endocrine therapy. When a brain metastasis was identified and surgically removed, this secondary tumour showed a dramatic change to ER-/PR-/HER2+ and a subtype switch to HER2-enriched (HER2E), along with a HER2 region amplification and an ERBB2/HER2 E668Q activating mutation.
The study found that, in nine out of 14 patients with both liver and lung metastases, liver metastases had lower immune cell infiltration than lung metastases. “This is important because the different sites of metastases in the same patient are likely to have a different response to immune checkpoint inhibitors, and a single biopsy of a liver metastasis would not be representative for treatment decisions in patients with more than one site of metastasis,” Garcia-Recio told Cancerworld.
In mouse models, the team is now exploring ways of reversing or treating the changes that they have observed. “One approach being investigated is to use demethylation agents in combination with immune checkpoint inhibitors to remove the methylation that is blocking the expression of the HLA-A genes,” says Charles Perou, the study’s corresponding author.
In the prospective part of the study, due to start soon, in addition to using the four genetic and genomic platforms, the US team plan to undertake panels of genetic mutations and collect blood samples in around 200 patients who have had metastases. Some of these results will be returned to the patient to guide treatment strategies.
Illustration from: https://www.youandbreastcancer.com/en-bc/view/m201-a02-understanding-metastatic-breast-cancer-animation