A pilot project pooling the procurement of cancer drugs has led to cancer institutes in India saving over USS 116 million – an 82% average reduction on the drugs’ reference prices.
The results of the pilot project, led by Tata Memorial Hospital in Mumbai and published in the Bulletin of World Health Organisation, show how 23 cancer centres which are part of India’s National Cancer Grid have pooled their purchase of 40 high-value cancer and supportive care drugs, normally worth US$197 million.
The pooled procurement process involved technical and financial evaluation, followed by contracts between individual centres and selected vendors.
Each participating centre made savings of between 23 and 99%. The greatest savings came with generic drugs rather than innovative and newly patented medicines. “This study reveals the advantages of group negotiation in pooled procurement for high-value medicines, an approach that can be applied to other health systems,” write the authors, led by Tata directors and researchers.
How it works
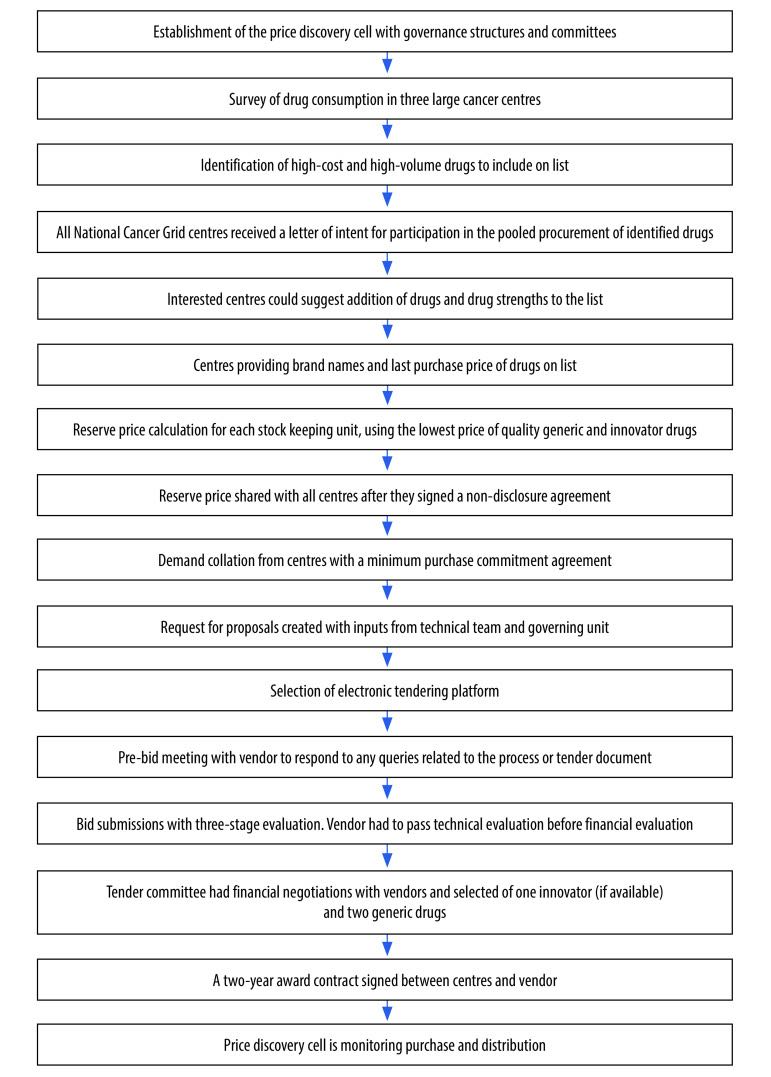
The process started with the formation of a ‘price discovery cell’, a group of experts tasked with assessing overall demand and negotiating prices down.
The cell started by conducting a pilot of three prominent Indian cancer centres to determine which drugs had the greatest impact on annual budgets. A full list of centres participating in the project was then finalised, after which the team collected data on demand for each drug and used projections to determine volumes required over the next two years.
The cell established the concept of “fair price” – the maximum price at which each participating member of the network would agree to purchase the drug. If the final price was higher than the fair price, the member could opt out of the process.
The team then used a tendering platform to submit tender forms and evaluate the tenders. Evaluation was done in three stages – prequalification, technical evaluation, and financial evaluation. Prequalification involved criteria such as whether the firm was compliant with WHO good manufacturing practices and was able to deliver to different locations. Technical evaluation involved assessing processes, transparency, compliance with regulation, and cold chain maintenance. Financial evaluation checked whether the price regulating authority had set the price for the drug, and whether that price was what the vendor had quoted to the pool.
Once this process was completed, the cell invited two suppliers with the lowest priced generic and innovative drugs into discussions, and finalised the price and conditions with the selected vendor. Finally, the centres signed agreements with the vendor for two years.
Prices were reduced for 40 drugs including cytotoxics, targeted therapies, antibiotics, antifungals, antiemetics and growth factors
Out of 46 vendors who participated in the process, 33 were selected for technical evaluation and 28 were selected for financial evaluation. Out of those, 24 were selected, out of which 21 agreed to supply the drugs at the pooled prices.
The process, from formation of the price discovery cell to signing of contracts, took one year to complete.
Prices were effectively reduced, both on reserve and maximum retail price, for 40 drugs used by cancer patients, including cytotoxic drugs, targeted therapies, antibiotics, antifungals, antiemetics and growth factors.
Potential impact
The success of this pilot has wide implications for health systems, as the most essential cancer drugs are not accessible in most low- and middle-income countries. Drugs account for approximately 40% of total cancer treatment costs and are increasingly unaffordable in low- to middle-income countries.

Even in high income countries, increasing costs of drugs is considered a hurdle to effective treatment. While there are numerous pooled procurement programmes in Europe, they are limited to a few drugs, and – unlike the Tata pilot’s approach – they often do not consider quality parameters.
The pilot project helped reduce drug prices for both big and small centres. For Tata Memorial Hospital, which has high volumes, the savings on maximum retail price increased from 55% to 85% thanks to the process, said medical oncologist Manju Sengar, co-author of the study.
The model also assures quality of drugs by using stringent parameters and addresses the issue of supply chain issues faced by smaller centres, Sengar said. Due to the pilot project’s success, 40 other centres and state health departments have expressed their interest in the second cycle, she said.
Ravi Mehrotra, Adjunct Professor, Rollins School of Public Health, USA and former Director of the National Institute of Cancer Prevention and Research, India, called the results of the pilot attempt “promising”. He said that, outside most major cancer hospitals in India, there are pharmacies which advertise huge discounts on cancer drugs, signalling huge margins on the drugs, but their quality may be questionable.
“If pooled procurement can ensure quality and steady supply at affordable prices, the idea will have a great impact on cancer treatment in India,” he said.
How it’s done: the Indian Cancer Grid approach to pooled procurement of cancer drugs
This is an open access article distributed under the terms of the Creative Commons Attribution IGO License (http://creativecommons.org/licenses/by/3.0/igo/legalcode), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. In any reproduction of this article there should not be any suggestion that WHO or this article endorse any specific organization or products. The use of the WHO logo is not permitted. This notice should be preserved along with the article’s original URL.
This article was amended to include the quote from Dr Ravi Mehrotra












